Progesterone Cream
This medication is compounded in Empower's PCAB-accredited 503A pharmacy outsourcing facility. Our quality process includes:
- Raw materials sourced from qualified, approved suppliers
- Finished product testing for potency, sterility, and endotoxins
- 100% visual inspection and AQL sampling - Certificate of Conformance generated for every batch
- Full batch review and release by our Quality Unit
- PCAB Accredited | FDA Registered | LegitScript Certified
Product Overview
Progesterone is one of several primary hormones in women that play a key role in the maintenance of pregnancy. After the rupture of the ovarian follicle and release of the ovum within the female ovary, progesterone is secreted by mature granulosa cells known as the corpus luteum. The released progesterone causes a thickening of the endometrial wall of the uterus, preparing it for the implantation of a fertilized ovum. If fertilization and implantation does not occur, the corpus luteum eventually atrophies and the serum progesterone levels fall. However, if implantation does occur, the serum progesterone levels remain elevated throughout the duration of the pregnancy as the placenta takes over the role of progesterone production.[1][2][3]
Progesterone is a derivative of cholesterol. After interaction with cellular membrane receptors and fusion with intracellular lysozymes, it is then transported to the mitochondria where it is then converted into pregnenolone. Through the mediating action of the Cytochrome P450 group of super enzymes, pregnenolone is then converted to progesterone within the corpus luteum or the placenta, depending on the presence or absence of pregnancy.[1]
In addition to its role in pregnancy, progesterone also plays an essential role in the central as well as peripheral nervous system. It is a neurosteroid, meaning that it is synthesized in the nervous system; this synthesis occurs in neuronal cells located with the central nervous system (CNS). Within the CNS, the pathway for progesterone synthesis is similar to that found in the corpus luteum or placenta. In the central nervous system, progesterone aids in the production of the myelin sheath, which is the layer surrounding the nerves, and is responsible for the transmission of rapid impulses along the nerve cells.[4]
The traditional means by which progesterone exerts its effects within the human body, also known as the genomic mechanism, is through its interaction with the progesterone receptor, PR. The progesterone receptor, typically described as a nuclear transcription factor, acts on ribosomal RNA to regulate ribosomal transcription, resulting in the production of the corresponding proteins. There are two primary forms of progesterone receptors that are located within the body namely PR-A and PR-B. PR-A and PR-B share the same DNA binding domain but have differing amino acid sequences with PR-A having 164 more amino acids than PR-B. Some of the effects of progesterone on organs within the body through its binding with the progesterone receptors PR-A and PR-B include:
Thickening of the uterine endometrium, getting it ready for the implantation of a fertilized ovum. Progesterone is released by the corpus luteum from the granulosa cells in the ovary for this purpose.
Maintenance of pregnancy, helping to ensure that a successfully implanted fertilized ovum is carried by the mother until delivery. After the development of the placenta, one of its roles is to continue the production of progesterone for the duration of the pregnancy, making sure that serum progesterone levels are sufficient to maintain the fetus within the uterus.[5]
In the female breast, progesterone stimulates the growth in number and size of the alveoli and lobes during pregnancy. In addition, progesterone inhibits the production of breast milk throughout the duration of the pregnancy. As progesterone levels fall after delivery, milk production within the breasts is triggered.
In the central nervous system, progesterone plays a role in the development of the myelin sheath surrounding the nerves. It has also been shown to play role in the neurological recovery from after a traumatic brain injury or a hypoxic-ischemic injury within the brain.[6]
In addition to the genomic mechanism, progesterone can also exert its effects through non-genomic means. The non-genomic pathway involves the activation of intracellular signaling pathways, which occurs through ion channels as well as second messenger cascades. This pathway normally occurs in the central nervous system; however, it can also take place in other areas of the body.[5]
There are certain circumstances under which progesterone should not be administered or, if administered, should be done with extreme caution. Some of the absolute or relative contraindications to progesterone administration include:
Hypersensitivity: Progesterone should not be administered to individuals who have a hypersensitivity to progesterone or any of its products.
Hepatic disease: Due to the fact that progesterone is mainly metabolized in the liver, its administration to individuals with significant renal impairment should be avoided as much as possible.
Ectopic pregnancy: Ectopic pregnancy as well as missed or incomplete abortions are absolute contraindications to exogenous progesterone administration.
Breastfeeding: Caution should be exercised when administering progesterone to breastfeeding mothers as detectable amounts have been found in breast milk.
Thromboembolic diseases: Individuals with a current or previous history of thromboembolic disease such as peripheral vascular disease, stroke, or myocardial infarction should not receive progesterone therapy. Individuals with high risk factors for thromboembolic diseases need to be closely monitored if they are given progesterone.
Vaginal bleeding: Progesterone should not be administered to females presenting with vaginal bleeding of an unknown cause.
Diabetes mellitus: Diabetics may receive progesterone therapy with caution as some individuals may experience a decrease in glucose tolerance and a resultant increase in blood sugar levels.
Reproductive cancer: Progesterone therapy is contraindicated in females presenting with breast, cervical, endometrial, ovarian, uterine, or vaginal cancer. Since these cancers are usually hormonally dependent, progesterone administration may worsen these conditions.
Hyperlipidemia: Progesterone therapy has been shown to impair lipid metabolism in some individuals and so should be administered cautiously to individuals with known or suspected cases of hyperlipidemia.
Drug interactions: Care should be taken when administering progesterone with other drugs due to the likelihood of significant drug interactions. Some drugs such as barbiturates can stimulate the production of Cytochrome P450 enzymes in the liver and increase the rate of hepatic clearance of progesterone; this can result in decreased serum levels of progesterone as well as decreased effectiveness.[9]
Some side effects that individuals may experience while on progesterone therapy are mild and generalized such as nausea and vomiting, urticaria, rhinitis, fever, headache, and fatigue, among others. Other individuals, however, may experience more severe and consequential adverse effects such as:
Thromboembolic disease: This is especially the case in individuals who have risk factors for thromboembolic disease.
Emotional lability: Sudden onsets of depression, anxiety, irritability, and uncontrollable outbursts are some emotional changes that may occur in women on progesterone therapy.
Fluid retention: Progesterone administration may cause fluid retention. In individuals with cardiac or renal disease, this may result in a worsening of their condition.
Menstrual changes: Changes in menstrual flow and rhythm, dysmenorrhea, and amenorrhea are some of the changes that may be seen in women receiving progesterone therapy.[9]
Progesterone is a US FDA pregnancy category B medication. Studies in animals have not demonstrated any particular risks to fetuses. However, enough adequate and well-controlled studies about its effects have not been done in pregnant women. Current recommendations are that progesterone therapy should only be used during the first three months of therapy. It should be discontinued once there is evidence of an adequate production of placental progesterone.[12]
Detectable levels of progesterone have been found in the milk of breastfeeding mothers. There have been no reported instances of developmental problems in neonates and toddlers of mothers with progesterone in their breast milk. As such, there are no restrictions for breastfeeding women receiving progesterone therapy. However, some degree of care should be exercised when administering progesterone to lactating females.
Store this medication in its original container at 68°F to 77°F (20°C to 25°C) and away from heat, moisture and light. Keep all medicine out of the reach of children. Throw away any unused medicine after the beyond use date. Do not flush unused medications or pour down a sink or drain.
- Taraborrelli, S., “Physiology, production and action of progesterone”, ACTA Obstetricia et Gynecologica, vol.94, pp. 8-16, 2015. Available: https://obgyn.onlinelibrary.wiley.com/doi/pdf/10.1111/aogs.12771
- Graham, J. D., Clarke, C.L., “Physiological Action of Progesterone in Target Tissues” Endocrine Reviews, vol.18, pp. 502-519, 1997. Available: https://academic.oup.com/edrv/article/18/4/502/2530774
- ”Progesterone”, National Center for Biotechnology information, 2020. Available: https://pubchem.ncbi.nlm.nih.gov/compound/Progesterone
- Schumacher, M., Hussain, R., Gago, N., Oudinet, J. P., Mattern, C., Ghoumari, A. M., “Progesterone synthesis in the nervous system: implications for myelination and myelin repair”, Frontiers in neuroscience, Vol.6, issue 10, 2012. Available: https://doi.org/10.3389/fnins.2012.00010
- Singh, M, Su, C, Ng, S., “Non-genomic mechanisms of progesterone action in the brain”, Frontiers in Neuroscience, vol.7, 2013. Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3776940/#:~:text=The%20%E2%80%9Cclassical%E2%80%9D%20mechanism%20by%20which,region%20of%20target%20genes%20to
- Gonzalez-Orozco, J.C., Camacho-Arroyo, I, “Progesterone Actions During Central Nervous System Development”, Frontiers in Neuroscience, vol.13, 2019. Available: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6533804/
- ”Progesterone - Drug summary”, Prescribers’ Digital Reference. Available: https://www.pdr.net/drug-summary/progesterone?druglabelid=2388
- ”Progesterone pregnancy and breastfeeding warnings” Available: https://www.drugs.com/pregnancy/progesterone.html#:~:text=US%20FDA%20pregnancy%20category%20B,controlled%20studies%20in%20pregnant%20women
Administration Instructions
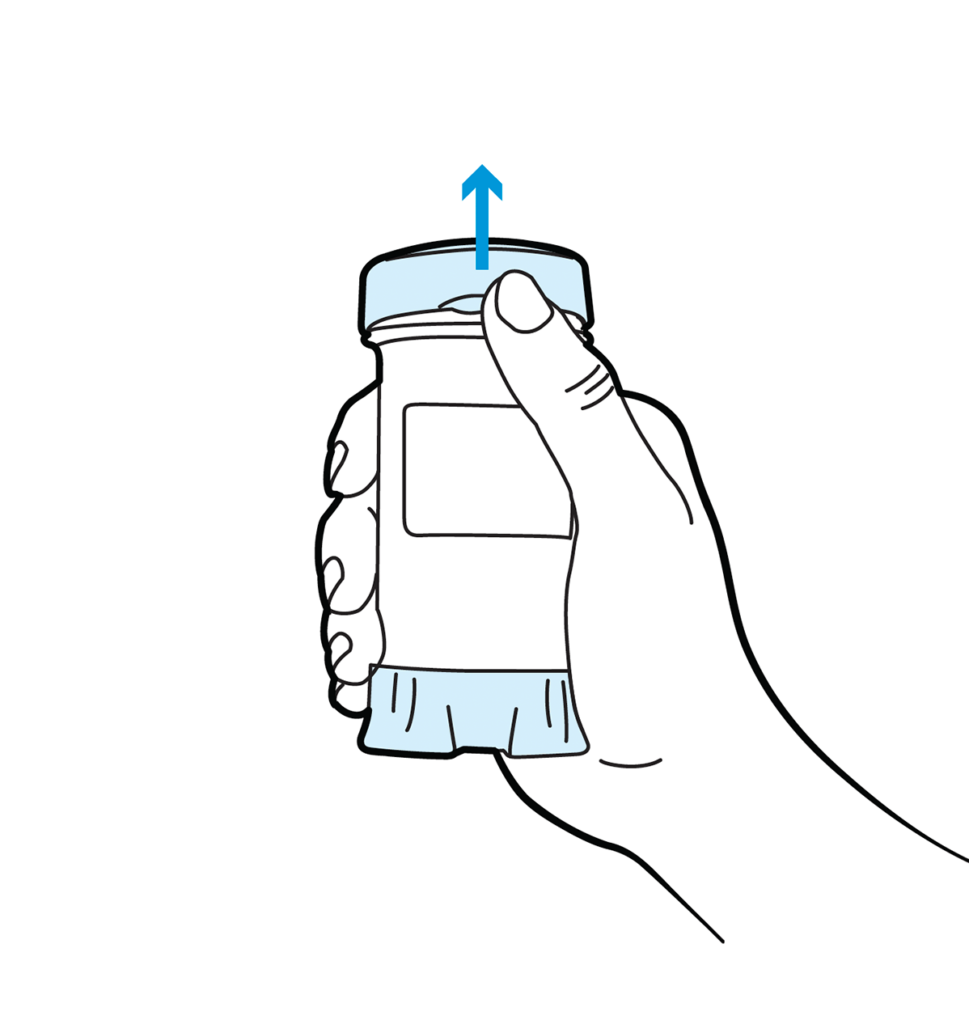
Topi-Click Dispensing Instructions
503A vs 503B
- 503A pharmacies compound products for specific patients whose prescriptions are sent by their healthcare provider.
- 503B outsourcing facilities compound products on a larger scale (bulk amounts) for healthcare providers to have on hand and administer to patients in their offices.
Frequently asked questions
Our team of experts has the answers you're looking for.
A clinical pharmacist cannot recommend a specific doctor. Because we are licensed in all 50 states*, we can accept prescriptions from many licensed prescribers if the prescription is written within their scope of practice and with a valid patient-practitioner relationship.
*Licensing is subject to change.
Each injectable IV product will have the osmolarity listed on the label located on the vial.

Given the vastness and uniqueness of individualized compounded formulations, it is impossible to list every potential compound we offer. To inquire if we currently carry or can compound your prescription, please fill out the form located on our Contact page or call us at (877) 562-8577.
We source all our medications and active pharmaceutical ingredients from FDA-registered suppliers and manufacturers.






 Progesterone Capsules
Progesterone Capsules Progesterone Troches
Progesterone Troches Progesterone Injection
Progesterone Injection Estradiol Cypionate Injection
Estradiol Cypionate Injection Bi-Est Cream
Bi-Est Cream Testosterone Cream
Testosterone Cream DHEA Capsules
DHEA Capsules DHEA / Pregnenolone Capsules
DHEA / Pregnenolone Capsules