Nandrolone Decanoate Injection
This medication is compounded in Empower's PCAB-accredited 503A pharmacy and/or FDA-registered 503B outsourcing facility. Our quality process includes:
- Raw materials sourced from qualified, approved suppliers
- Finished product testing for potency, sterility, and endotoxins
- 100% visual inspection and AQL sampling - Certificate of Conformance generated for every batch
- Full batch review and release by our Quality Unit
- PCAB Accredited | FDA Registered | LegitScript Certified
Product Overview
Nandrolone Decanoate is a compounded, prescription-only anabolic steroid injection. It is a synthetic derivative of testosterone (specifically a 19-nortestosterone ester) with pronounced anabolic properties and moderate androgenic activity.[1][5]
Clinically, nandrolone decanoate has been used to manage certain forms of anemia (particularly anemia of chronic kidney disease) and to promote tissue building in debilitating conditions. For example, it may help increase red blood cell production and lean muscle mass in patients with chronic illness or wasting syndromes such as HIV/AIDS, and it has even been employed historically in osteoporosis (in postmenopausal women) and some cases of advanced breast cancer.[1][5]
Due to its potential for misuse as a performance-enhancing drug, nandrolone decanoate is classified as a Schedule III controlled substance in the United States. This compounded formulation is not commercially available as a manufactured drug product in the U.S., and is instead prepared by compounding pharmacies and outsourcing facilities.[1][5]
Given its potent physiological effects and risk profile, therapy with nandrolone decanoate is initiated and monitored only by qualified healthcare professionals. Patients receiving this medication are closely supervised to maximize therapeutic benefits while minimizing adverse effects.[1][5]
Nandrolone decanoate is administered by deep intramuscular (IM) injection, typically into a large muscle such as the gluteus (buttock) muscle. Injections are usually given by a healthcare professional, though in some cases patients or caregivers trained in proper IM technique may administer the doses. Dosage must be individualized based on the patient’s condition, response to therapy, and tolerability. As this medication may be used in a variety of settings (from anemia management to adjunct therapy in catabolic states), there is no single fixed dose for all patients. However, dosing guidelines can be drawn from clinical experience and literature.[1][2]
For instance, in the treatment of anemia of chronic renal failure, a common dosage regimen has been 100-200 mg of nandrolone decanoate per week in male patients, and about 50-100 mg per week in female patients. Therapy is usually considered adjunctive to other treatments (e.g. adequate iron supplementation in anemia). If no hematologic or clinical improvement is observed within about 6 months, discontinuation should be considered. In other scenarios, such as cachexia or osteoporosis, doses in the range of ~50-200 mg administered every 1-4 weeks have been used, depending on the severity of the condition and patient factors. It is often recommended that anabolic steroid therapy be intermittent if possible, for example, treatment cycles with drug holidays, to reduce the risk of long-term adverse effects. The duration of therapy will depend on the therapeutic goals and patient response; some patients may receive a short course for a few months, while others (such as those with chronic anemia) might be treated longer under careful supervision. In all cases, the benefit-to-risk ratio should be regularly assessed by the prescribing provider. If significant adverse effects emerge, dose reduction or drug discontinuation may be necessary.[1][2]
For pediatric patients, nandrolone decanoate use is very limited. When used (for example, in certain severe anemias in children or for constitutional growth failure in rare cases), doses are much lower and infrequent. The literature suggests pediatric dosing on the order of 25-50 mg IM every 3-4 weeks in children aged 2 to 13 years, although safety and efficacy in pediatric patients have not been firmly established. Extreme caution is required in pediatric use because of the risk of accelerating epiphyseal closure. Regular monitoring of bone growth via X-ray every 6 months is recommended if a child or adolescent is undergoing nandrolone therapy. If any compromise in expected growth is detected, therapy should be re-evaluated and likely discontinued to prevent permanent stunting.[1][2]
Nandrolone decanoate is compounded in a sterile oil-based solution (using a grapeseed oil vehicle) at a concentration of 200 mg/mL, and in this formulation it is supplied in multi-dose vials (5 mL vials containing a total of 1000 mg of nandrolone decanoate). Each milliliter of solution also contains a preservative (such as benzyl alcohol) to maintain sterility in a multi-use vial. Before administration, the solution should be inspected visually for any particulate matter or discoloration. If the solution has crystallized or shows signs of contamination, it must not be used. The injection should be given using aseptic technique into the muscle tissue; care should be taken to avoid intravascular injection. Rotating injection sites is advisable if repeat injections are needed over long periods. After withdrawing the dose, any unused solution in the vial should be stored properly (see Storage & Handling) and used within the designated beyond-use date provided by the pharmacy.[3]
During therapy, monitoring parameters will typically include the patient’s blood counts (to gauge efficacy in anemia and watch for polycythemia), liver function tests, lipid profile, and hormone levels (testosterone, etc.). Depending on the indication, other measures like bone density or nitrogen balance may be tracked. Dose adjustments should be made based on these monitoring results and the clinical response. In general, therapy with nandrolone decanoate is approached as an adjunct to supportive care (nutritional and otherwise) rather than a standalone solution. Adequate protein and calorie intake is necessary to maximize the anabolic effects; for example, in patients with anemia or wasting, addressing underlying nutritional deficiencies is crucial for nandrolone to be effective. By following appropriate dosing guidelines and closely monitoring the patient, healthcare providers aim to use the lowest dose that achieves the desired clinical outcome, thereby reducing the risk of adverse reactions.[3]
Nandrolone decanoate is an intramuscular depot form of nandrolone, acting as a prodrug that slowly releases nandrolone into circulation. Nandrolone is an agonist of the androgen receptor (AR) in target tissues, which may lead to increased protein synthesis, enhanced nitrogen retention in muscle, and stimulation of erythropoiesis (red blood cell production). These actions underlie its anabolic effects, such as promoting muscle growth, bone density, and red cell mass, which may be beneficial in catabolic states.[1][5]
Compared to endogenous testosterone, nandrolone exhibits a higher ratio of anabolic-to-androgenic activity, meaning it tends to favor muscle and bone-building effects over masculinizing effects. This pharmacologic profile is partly due to nandrolone’s unique metabolism: it is less readily converted to the potent androgen dihydrotestosterone (DHT) in androgen-sensitive tissues, resulting in relatively reduced AR stimulation in tissues like skin and prostate. Instead, 5α-reductase enzymes convert nandrolone to a weaker androgen (dihydronandrolone), which may contribute to a lower incidence of certain androgenic side effects (e.g. scalp hair loss or prostate enlargement) as compared to testosterone.[1][5]
Nevertheless, complete dissociation of anabolic and androgenic effects has not been achieved; nandrolone can still cause androgenic effects in the body. It may suppresses the hypothalamic-pituitary-gonadal axis via negative feedback, leading to reduced gonadotropin secretion and a consequent decline in endogenous testosterone and sperm production.[1][5]
The decanoate ester confers a prolonged duration of action: following intramuscular injection, nandrolone decanoate is slowly absorbed and hydrolyzed, with an effective half-life of about 6-12 days and activity that may persist for 2-3 weeks. Pharmacologically, it has minimal estrogenic activity (it can aromatize to estradiol to a small extent) and does possess some progestogenic affinity, which together can contribute to effects like fluid retention and changes in lipid metabolism.[1][5]
In summary, nandrolone decanoate’s mechanism is similar to that of other anabolic steroids, binding AR to exert tissue-building effects, but its kinetic profile and metabolism grant it a distinctive balance of potent anabolic efficacy with comparatively reduced (but not absent) androgenic and estrogenic side effects. All such effects are dose-dependent, and higher doses or prolonged use will increase the likelihood of androgenic and metabolic side effects.[1][5]
Nandrolone decanoate is contraindicated in patients with hormone-sensitive malignancies and in certain other conditions where androgenic therapy would pose undue risk. This includes men with carcinoma of the breast or known or suspected prostate carcinoma, as exogenous androgens may stimulate the growth of these tumors.[1][4]
It is also contraindicated in women with breast cancer who have hypercalcemia, since anabolic steroids can induce osteolytic bone resorption and worsen hypercalcemia. Use is strictly contraindicated during pregnancy, as androgens like nandrolone can cause virilization of the external genitalia of a female fetus and other fetal harm.[1][4]
Patients with nephrosis or nephrotic phase of nephritis (severe renal disease) should not receive nandrolone decanoate, because anabolic steroids can cause fluid retention that may exacerbate edema in this population. Furthermore, nandrolone decanoate is generally avoided in individuals with severe hepatic impairment or active cardiac disease (such as advanced congestive heart failure) due to the risk of fluid retention and cholestatic complications that androgens may precipitate.[1][4]
In addition, patients with known hypersensitivity to nandrolone or any of the formulation components (e.g. the oil base or benzyl alcohol preservative) should not use this preparation. Extreme caution or avoidance is advised in pediatric patients who have not yet completed growth, since anabolic steroids can prematurely close epiphyseal growth plates (see Pediatric Use).[1][4]
Overall, nandrolone decanoate should only be prescribed after a thorough evaluation, and it is contraindicated in any situation where the risks of androgen therapy are deemed to outweigh potential benefits. This list may not be exhaustive, and prescribers will consider all comorbid conditions before initiating therapy.[1][4]
Androgens like nandrolone can interact with other medications and may also alter certain laboratory test results.
Anticoagulants: Anabolic steroid therapy can enhance the anticoagulant effects of oral anticoagulants (such as warfarin) by mechanisms that are not fully understood, potentially increasing sensitivity to these drugs. Patients on warfarin or similar anticoagulants may require closer monitoring and dose adjustments of the anticoagulant when nandrolone is added or discontinued. Coagulation parameters (e.g. prothrombin time/INR) should be checked frequently, as nandrolone has been reported to prolong prothrombin time in some cases.[1][5]
Hormonal agents: Drugs that affect androgen signaling may theoretically influence the activity of nandrolone. For instance, concurrent use of 5α-reductase inhibitors (like finasteride or dutasteride, used for prostate conditions) could alter nandrolone’s metabolism and reduce its conversion to less active metabolites, potentially modifying its androgenic effects. Likewise, the use of other steroid hormones or hormone-modulating supplements can have unanticipated additive or opposing effects; an example is saw palmetto (Serenoa repens), an herbal product with anti-androgenic properties, which might counteract some actions of anabolic steroids if taken together.[1][5]
Adrenal steroids: Concomitant administration of corticosteroids or ACTH with nandrolone may increase the risk of edema and fluid retention. This is particularly important in patients with cardiac, renal, or hepatic disease, as combined glucocorticoid and androgen therapy can synergistically promote sodium retention and swelling in these vulnerable individuals.[1][5]
Diabetes medications: Anabolic steroids can affect carbohydrate metabolism. Nandrolone may influence blood glucose levels by improving insulin sensitivity in some hypogonadal individuals or, conversely, by reducing glucose tolerance in others. Diabetic patients starting nandrolone should be observed for changes in glycemic control, as adjustments in anti-diabetic medication dosing could be required.[1][5]
Thyroid function tests: Nandrolone decanoate can decrease the levels of thyroxine-binding globulin (TBG) in the bloodstream, leading to a reduction in total T₄ (thyroxine) levels and an increase in resin uptake of T₃/T₄ on assays. Importantly, free thyroid hormone levels and clinical thyroid status remain unchanged; in other words, nandrolone may alter laboratory values without causing true hypothyroidism. Clinicians interpreting thyroid labs in patients on anabolic steroids should be aware of this artifact.[1][5]
In summary, patients should inform their healthcare provider of all concomitant medications, including over-the-counter drugs and supplements, before starting nandrolone decanoate. This helps to avoid and manage potential drug interactions. Regular monitoring of blood work is also advisable to detect changes in lipid profile, liver enzymes, blood count, or other parameters during therapy (since androgens can impact these values).[1][5]
Like all anabolic steroids, nandrolone decanoate is associated with a broad range of potential adverse effects involving multiple organ systems. The occurrence and severity of side effects depend on the dose, duration of use, and patient-specific factors (such as sex and age).[1][6]
Endocrine and Reproductive: In men, nandrolone may suppress the normal hypothalamic-pituitary-gonadal axis, leading to inhibition of testicular function with resultant testicular atrophy and reduced sperm count (oligospermia). Decreased endogenous testosterone production can cause infertility while on therapy, which in most cases may be reversible after discontinuation over time. Male patients may also experience gynecomastia (benign breast tissue enlargement) due to steroid-induced hormonal imbalances. Prolonged or high-dose use in men may carry a risk of chronic priapism (prolonged erections), impotence/erectile dysfunction, and epididymal inflammation.[1][6]
In women, even at therapeutic doses, the most common adverse effect is virilization, the development of male secondary sexual characteristics. Signs of virilization in female patients can include acne, hirsutism (excessive body and facial hair growth), male-pattern baldness, deepening of the voice, increased libido, menstrual irregularities (oligomenorrhea or amenorrhea), and clitoromegaly (enlargement of the clitoris). Many of these masculinizing changes, especially voice deepening and clitoral enlargement, may be irreversible or only partially reversible upon discontinuing the drug. Therefore, female patients require careful monitoring, and if virilization signs emerge, therapy is usually halted promptly to limit permanency.[1][6]
In both sexes, anabolic steroids can cause either an increase or decrease in libido, and mood changes related to hormonal effects are not uncommon.[1][6]
Dermatologic: Androgenic stimulation can lead to sebaceous gland hypertrophy and acne, particularly in women and adolescent males. Oily skin and hair, and predisposition to male-pattern hair loss (alopecia) in those genetically susceptible, have been reported.[1][6]
Psychiatric/CNS: Nandrolone and other AAS may affect mood and behavior. Some individuals experience mood swings, including irritability, aggression, or depression. Patients might report insomnia or changes in mental focus as well. There is a potential for habituation or psychological dependence with anabolic steroids, especially when misused at high doses, leading to withdrawal symptoms upon cessation.[1][6]
Cardiovascular and Metabolic: Nandrolone decanoate may cause fluid retention and edema due to its effects on electrolyte balance (promoting sodium and water retention). This can manifest as swelling in the extremities and, in predisposed individuals, may contribute to or worsen hypertension. Anabolic steroid use has been linked to adverse lipid changes: nandrolone may increase LDL cholesterol and decrease HDL cholesterol levels, potentially accelerating atherogenic risk.[1][6]
Prolonged use or abuse of AAS, even in the absence of other risk factors, has been associated with cardiovascular complications including cardiomyopathy, arrhythmias, and heart failure. Indeed, cases of congestive heart failure and dilated cardiomyopathy have been reported in patients with a history of high-dose steroid abuse, though such severe outcomes are typically seen with supra-physiologic dosages. Even at therapeutic doses, caution is warranted in those with underlying heart disease, and any signs of dyspnea, edema, or chest pain should prompt evaluation.[1][6]
Hematologic: By stimulating erythropoiesis, nandrolone can increase red blood cell mass; in some patients this may lead to polycythemia (elevated hematocrit) which can increase blood viscosity and risk of thrombosis if not monitored. Paradoxically, in patients on anticoagulants, the enhanced clotting factor suppression by androgens can increase bleeding tendency (as noted in Interactions).[1][6]
Hepatic: Unlike oral 17α-alkylated anabolic steroids, nandrolone (being parenteral and not 17α-alkylated) is not commonly associated with acute liver toxicity such as cholestatic jaundice at normal doses. However, serious hepatic adverse effects have been documented with long-term anabolic steroid therapy. Peliosis hepatis, a condition where blood-filled cysts form in the liver, has been reported in patients receiving chronic androgenic steroid treatment. These cysts may be asymptomatic until they cause liver dysfunction or rupture; they have occasionally led to life-threatening intra-abdominal hemorrhage.[1][6]
Hepatocellular neoplasms (liver tumors) have also been observed with extended anabolic steroid use; many are benign adenomas that may regress after drug withdrawal, but malignant liver tumors have been reported in rare cases. Regular monitoring of liver function tests is advised during prolonged therapy.[1][6]
Musculoskeletal: In pediatric patients, anabolic steroids can accelerate epiphyseal closure in the long bones, leading to premature cessation of growth. This effect can result in a permanently reduced adult stature if not detected early; thus, any use of nandrolone in children requires periodic X-ray monitoring of bone age. In adults, supraphysiologic doses of AAS have been linked to tendon injuries, possibly due to disproportionate muscle strength gains outpacing tendon adaptation.[1][6]
Other effects: Nandrolone can cause voice changes (deepening) in women, which may be irreversible. Some users report headaches, nausea, or gastrointestinal discomfort such as diarrhea with anabolic therapy. Changes in glucose tolerance or even mild insulin resistance have been noted in some individuals. Injection-site reactions (pain, redness, or swelling) can occur, though injection-site abscesses are rare when proper technique is used. Because nandrolone decanoate is typically administered in a sesame oil vehicle, allergic reactions are uncommon but possible in those with sesame oil sensitivity.[1][6]
Overall, the safety profile of nandrolone decanoate requires vigilant monitoring. Many side effects are dose-dependent and may be mitigated by using the lowest effective dose for the shortest necessary duration. Some adverse effects (like lipid changes or mild hepatic enzyme elevations) are reversible upon discontinuation, whereas others (virilization, growth stunting, etc.) can leave lasting effects. Patients should be educated on recognizing early signs of serious side effects (such as excessive virilization, jaundice, or edema) and advised to report them promptly. Healthcare providers will periodically evaluate blood counts, liver enzymes, lipid panels, and hormonal levels during therapy to ensure patient safety. By closely supervising therapy and individualizing dosing, practitioners aim to harness the anabolic benefits of nandrolone decanoate while minimizing its risks.[1][6]
Pregnancy: Nandrolone decanoate is contraindicated for use during pregnancy (FDA Pregnancy Category X). Anabolic steroids can cause severe adverse effects on the fetus. In particular, exposure to nandrolone or other androgens in utero, especially during the first trimester, may lead to virilization of a female fetus; for example, development of male-like genitalia in a genetically female fetus. There are no controlled studies of nandrolone in pregnant women because it would be unethical to expose a fetus to this drug; however, animal studies and the pharmacology of the drug strongly indicate risk of teratogenicity and fetal harm.[1][4]
Women who are pregnant or who may become pregnant must not use nandrolone decanoate. If a patient becomes pregnant while receiving nandrolone, the medication should be discontinued immediately and the patient apprised of the potential hazard to the fetus.[1][4]
Lactation: It is unknown whether nandrolone or its metabolites are excreted into human breast milk. Given that many drugs are excreted in breast milk and that anabolic steroids could potentially cause serious adverse effects in a nursing infant, women who are breastfeeding are generally advised not to use nandrolone decanoate.[1][4]
If an anabolic steroid is absolutely indicated in a postpartum patient, a decision should be made to discontinue nursing or avoid the drug altogether, weighing the importance of treatment to the mother. In practice, because nandrolone is usually an elective therapy (for conditions that have alternative treatments or can be deferred), lactating patients are counseled to use other options or to postpone nandrolone until breastfeeding has ceased. Additionally, anabolic steroids may suppress lactation or alter the composition of breast milk due to their hormonal effects, which provides further rationale to avoid these agents during nursing.[1][4]
Contraception: Women of childbearing potential on nandrolone should use effective contraception to prevent pregnancy during therapy.[1][4]
Fertility considerations: Although not directly a pregnancy or lactation issue, it is worth noting that nandrolone can impair fertility in male patients by inducing reversible oligospermia or azoospermia with prolonged use. Men desiring to father a child should be aware that anabolic steroid use may need to be halted well in advance of attempting conception to allow recovery of sperm production.[1][4]
In summary, nandrolone decanoate should not be used in pregnancy or while breastfeeding. Any woman who becomes pregnant while on nandrolone must discontinue it, and nursing mothers are advised to avoid this medication to protect the infant.[1][4]
Proper storage and handling of nandrolone decanoate injection are important to maintain its stability and sterility. The multi-dose vials should be stored at controlled room temperature, typically around 20°-25°C (68°-77°F). Excursions outside this range should be minimized; the compound should not be frozen, and it is best kept in a cool, dry place. The vials must be protected from light. Nandrolone decanoate solution is light-sensitive, and exposure to direct light could potentially degrade the active ingredient over time. For this reason, the medication is often dispensed in amber-colored vials or kept within its carton until ready for use. Users should keep the vial in its original packaging when not in use and store it away from sunlight or strong artificial light.[1][2]
Because this is a compounded sterile preparation, it will have a beyond-use date (BUD) assigned by the pharmacy rather than a manufacturer’s expiration date. The BUD indicates how long the vial’s contents remain stable and sterile under recommended storage conditions. Patients should note the BUD on the label and not use the medication beyond that date. Once the BUD or expiration is reached, any unused solution should be disposed of properly.
The vial should be stored out of reach of children and pets for safety. When handling the vial, users should inspect it for any cracks or damage before each use, as compromised vials could lead to contamination. Syringes and needles used to withdraw and inject the solution should be sterile and preferably used only once (needles should never be re-entered into the vial with a non-sterile needle). If multiple doses are drawn from the same vial over time, the rubber stopper should be swabbed with an appropriate antiseptic (e.g. isopropyl alcohol) before each puncture to maintain sterility.
If the solution appears cloudy, has precipitated (crystals), or shows particles, do not use it and contact the pharmacy. Nandrolone decanoate in solution is usually a clear to yellowish oily liquid; any marked change in appearance could indicate contamination or instability. Likewise, if a vial develops an unusual odor or if there is any suspicion it may have been contaminated or stored improperly, it should be discarded according to medical waste protocols.
Disposal of unused or expired anabolic steroid medication should follow state and federal guidelines for controlled substances. Patients should not simply throw vials in the trash or flush them. Instead, return programs or consultation with the pharmacy for safe disposal methods are recommended due to the medication’s hormonal activity and controlled status.
In summary, store nandrolone decanoate vials at room temperature in a light-protected environment, observe the beyond-use date, and handle the vial with sterile technique. By adhering to these storage and handling recommendations, the potency and sterility of the medication are maintained for the duration of its use.
- Cleveland Clinic. (n.d.). Nandrolone. In Cleveland Clinic Health Library. Retrieved May 3 2025, from https://my.clevelandclinic.org/health/drugs/24940-nandrolone
- Hassan, D. A. E., Imam, H., Zidan, N., et al. (2023). The toxic effects of anabolic steroids “nandrolone decanoate” on cardiac and skeletal muscles with potential ameliorative effects of silymarin and fenugreek in rats. BMC Pharmacology and Toxicology, 24(1), 17. https://bmcpharmacoltoxicol.biomedcentral.com/articles/10.1186/s40360-023-00658-x
- Patanè, F. G., Liberto, A., Maglitto, A. N. M., et al. (2020). Nandrolone decanoate: Use, abuse and side effects. Medicina (Kaunas), 56(11), 606. https://doi.org/10.3390/medicina56110606
- Kicman, A. T. (2008). Pharmacology of anabolic steroids. British Journal of Pharmacology, 154(3), 502-521. https://doi.org/10.1038/bjp.2008.165
- American Regent, Inc. (2010). Nandrolone Decanoate Injection, USP - Package Insert. Shirley, NY: American Regent Pharmaceuticals. Retrieved Mar 25 2025, from https://www.drugs.com/pro/nandrolone.html
What is Nandrolone Decanoate and why is it compounded?
Nandrolone Decanoate is a synthetic anabolic steroid similar to testosterone, used for its muscle-building and red blood cell boosting effects. It is provided as an intramuscular injection. In the U.S., no mass-manufactured version is currently marketed, even though it was FDA-approved in the past.[1][2]
Therefore, compounding pharmacies and outsourcing facilities prepare this medication on a prescription basis to ensure patients who need it can obtain it. Compounding allows flexibility in concentration and volume, for instance, producing a 5 mL vial at 200 mg/mL, which can be useful for tailoring doses. This medication is prescription-only and not available as an over-the-counter supplement.[1][2]
Importantly, while compounded nandrolone is made in licensed pharmacies and outsourcing facilities following quality guidelines, it is not evaluated by the FDA for safety or efficacy in the way an FDA-approved drug is. Its use is reserved for cases where a physician determines the benefits for a specific patient outweigh the risks, and no suitable approved alternative is available.[1][2]
In summary, nandrolone decanoate is compounded to meet individual patient needs in a manner that commercial products currently do not, with oversight by the prescribing practitioner and compounding pharmacist to ensure quality and patient safety.[1][2]
What conditions might a doctor prescribe Nandrolone Decanoate for?
Doctors may prescribe nandrolone decanoate for a few specific medical situations, primarily when its anabolic properties can provide therapeutic benefit. The most common FDA-approved indication historically was anemia associated with chronic kidney failure, where nandrolone can stimulate red blood cell production.[3]
It has also been used off-label in managing HIV/AIDS wasting syndrome to help increase lean body mass and weight, in severe burns or chronic wound-healing situations to promote tissue rebuilding, and in certain cases of osteoporosis (particularly in postmenopausal women) to improve bone density. Additionally, nandrolone (or similar anabolic steroids) was used in the past as an adjunct treatment in advanced breast cancer for its tissue-building and appetite-stimulating effects, although this is less common now with newer therapies.[3]
It’s important to note that these uses are carefully weighed by physicians, for example, in cachexia (wasting conditions) or refractory anemia, a doctor might consider nandrolone if standard treatments are insufficient. Nandrolone is not a first-line therapy for any condition today; it’s typically reserved for specific cases such as anemia of renal disease or cachexia where other interventions haven’t achieved desired results.[3]
The prescription of nandrolone decanoate is individualized; two patients with the same diagnosis might not both be candidates for nandrolone, depending on their overall health and risk factors. Because of its side effect profile, this steroid is only used when the potential functional improvements (in muscle strength, weight, bone mass, etc.) are deemed to meaningfully enhance the patient’s quality of life or recovery course under close supervision.[3]
Always, the decision to use nandrolone is made by a qualified healthcare provider who can monitor the patient’s response and adjust treatment as needed.[3]
How long does it take to see results with Nandrolone Decanoate?
The onset of benefits from nandrolone decanoate can vary depending on the condition being treated and the individual patient. Generally, because this is a long-acting depot injection, it does not produce immediate effects; rather, its action unfolds over weeks. In anemia of chronic disease, improvements in hemoglobin might be observed within 4-6 weeks of consistent therapy if the drug is effective. Patients often require a few cycles of treatment (several injections) before significant increases in red blood cell indices are measurable.
For muscle wasting or strength, it may take a couple of months to notice a meaningful difference in weight or muscle mass, especially when combined with nutrition and exercise. Clinical trials of anabolic steroids in cachectic patients have noted gains in lean body mass typically after 8-12 weeks of therapy, although some patients report improved appetite or endurance sooner.
In osteoporosis, changes in bone density are very gradual; one would not “feel” a difference in bones, but over 6 months to a year, bone density scans might show modest increases if nandrolone is effective. It is important to set realistic expectations: nandrolone is not a fast-acting drug. Because it is administered every 1-4 weeks, its effects build up cumulatively. Also, individual response varies; some patients may respond quicker (or more robustly) than others.
The healthcare provider will typically assess intermediate markers (like weight, muscle circumference, blood counts) every few weeks to gauge if the patient is trending in the desired direction. If no response is evident after a trial period (often about 3-6 months, depending on the indication), the provider may decide to discontinue therapy. Conversely, if good improvement is seen, they might continue the course but will still usually institute a planned endpoint or at least a reassessment point to prevent indefinite use.
Patients should maintain close communication with their provider during this time and continue supportive measures (diet, physical therapy, etc.), as nandrolone’s benefits are maximized when part of a comprehensive treatment plan.
Administration Instructions
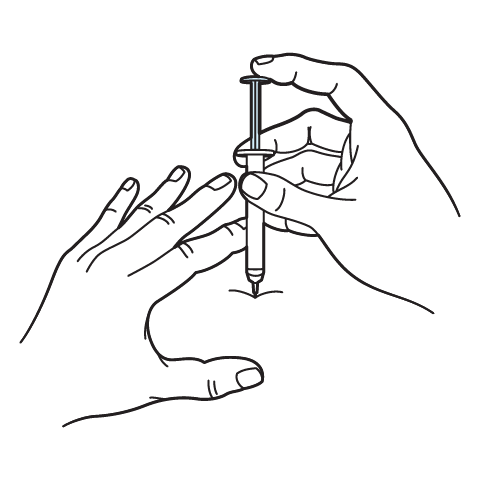
Intramuscular Injection Instructions
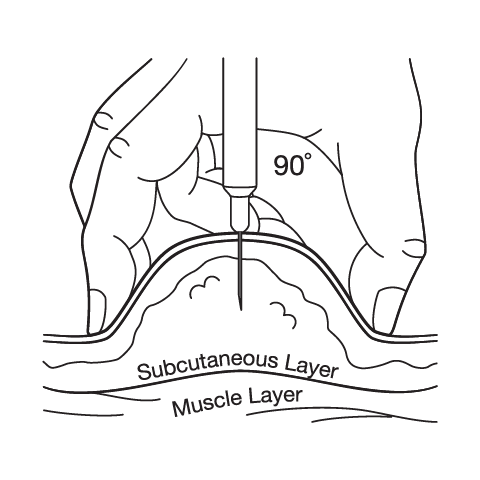
Subcutaneous Injection Instructions
503A vs 503B
- 503A pharmacies compound products for specific patients whose prescriptions are sent by their healthcare provider.
- 503B outsourcing facilities compound products on a larger scale (bulk amounts) for healthcare providers to have on hand and administer to patients in their offices.
Frequently asked questions
Our team of experts has the answers you're looking for.
A clinical pharmacist cannot recommend a specific doctor. Because we are licensed in all 50 states*, we can accept prescriptions from many licensed prescribers if the prescription is written within their scope of practice and with a valid patient-practitioner relationship.
*Licensing is subject to change.
Each injectable IV product will have the osmolarity listed on the label located on the vial.

Given the vastness and uniqueness of individualized compounded formulations, it is impossible to list every potential compound we offer. To inquire if we currently carry or can compound your prescription, please fill out the form located on our Contact page or call us at (877) 562-8577.
We source all our medications and active pharmaceutical ingredients from FDA-registered suppliers and manufacturers.


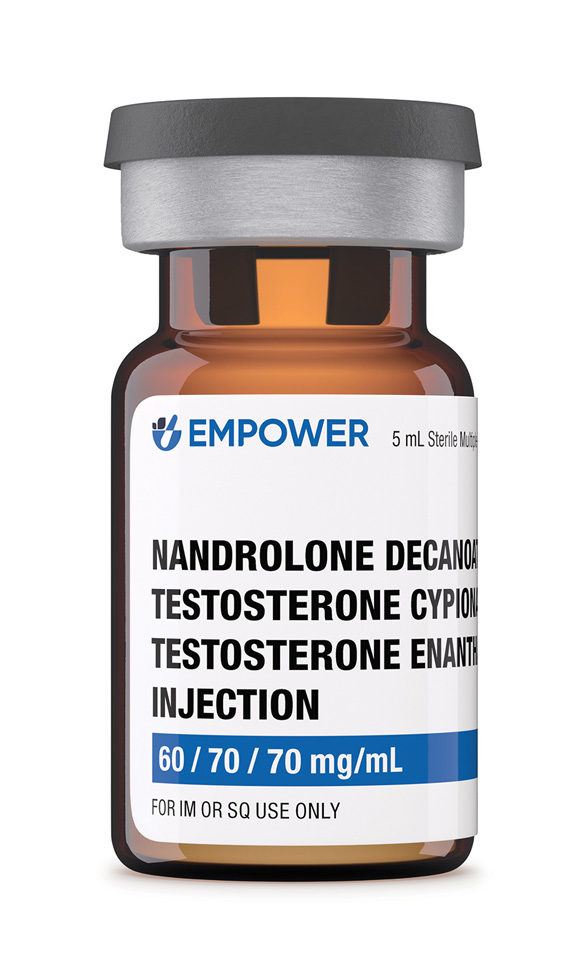 Nandrolone Decanoate / Testosterone Cypionate / Testosterone Enanthate Injection
Nandrolone Decanoate / Testosterone Cypionate / Testosterone Enanthate Injection Oxandrolone Tablets
Oxandrolone Tablets Oxandrolone Troches
Oxandrolone Troches Testosterone Cypionate / Testosterone Propionate Injection
Testosterone Cypionate / Testosterone Propionate Injection Anastrozole Capsules
Anastrozole Capsules Enclomiphene Citrate Capsules
Enclomiphene Citrate Capsules HCG Injection
HCG Injection Sermorelin Acetate Injection
Sermorelin Acetate Injection