Prednisone Tablets
† commercial product
This product is available solely through our 503A Compounding Pharmacy, ensuring personalized care and precision in every order. Please note that a valid prescription is required for purchase. If you do not have an account, please contact us.
Product Overview
Prednisone tablets represent a synthetic corticosteroid medication that may serve as an anti-inflammatory and immunosuppressive agent in various clinical applications. Prednisone is available in a 5 mg tablet strength for precise dosing flexibility.[1] Prednisone belongs to the glucocorticoid class of steroid hormones, which are structurally similar to cortisol, a hormone naturally produced by the adrenal cortex.[2] The medication may be prescribed for a wide range of inflammatory, autoimmune, and allergic conditions where corticosteroid therapy could provide therapeutic benefit.[3]
The pharmaceutical formulation of prednisone tablets involves careful consideration of bioavailability and pharmacokinetic properties to ensure consistent therapeutic outcomes.[4] Healthcare providers may consider prednisone therapy for patients experiencing conditions that could benefit from anti-inflammatory or immunosuppressive intervention.[5] The 5 mg tablet strength allows for flexible dosing regimens that can be tailored to individual patient needs and specific clinical circumstances.[6] Prednisone may undergo hepatic conversion to prednisolone, which represents the active metabolite responsible for the medication’s therapeutic effects.[7]
Clinical applications of prednisone may encompass a broad spectrum of medical conditions, including but not limited to inflammatory disorders, autoimmune diseases, allergic reactions, and certain malignancies.[8] The medication’s versatility in treating diverse pathological processes stems from its ability to modulate inflammatory pathways and immune system responses.[9] Healthcare professionals may prescribe prednisone as part of comprehensive treatment strategies that could include other medications and supportive care measures.[10] Patient selection for prednisone therapy typically involves careful evaluation of potential benefits versus risks, considering individual patient factors and underlying health conditions.[11]
The oral route of administration offers several advantages, including ease of use, patient convenience, and predictable absorption characteristics.[12] Prednisone tablets may be administered with or without food, though taking the medication with meals could help minimize potential gastrointestinal side effects.[13] The relatively long half-life of prednisone and its active metabolite prednisolone may allow for once-daily or divided dosing regimens, depending on the specific indication and patient response.[14] Clinical monitoring during prednisone therapy may include regular assessment of therapeutic response, adverse effects, and laboratory parameters that could be affected by corticosteroid treatment.[15]
Prednisone dosing must be individualized based on the specific condition being treated, disease severity, patient response, and tolerance to therapy.[111] The 5 mg tablet strength provides flexibility for precise dose titration and may be particularly useful for pediatric patients or adults requiring lower doses.[112] Initial dosing strategies may vary considerably depending on the clinical indication, ranging from low-dose anti-inflammatory therapy to high-dose immunosuppressive regimens.[113] Healthcare providers should consider the patient’s previous corticosteroid exposure, concurrent medications, and underlying health conditions when determining appropriate dosing.[114]
For many inflammatory conditions, initial doses may range from 5 to 60 mg daily, depending on the severity of the condition and the urgency of treatment required.[115] Acute severe conditions may require higher initial doses, while chronic conditions may be managed with lower maintenance doses.[116] The concept of pulse therapy involves using high doses for short periods followed by dose reduction or drug-free intervals.[117] This approach may help minimize long-term adverse effects while maintaining therapeutic efficacy for certain conditions.[118]
Dose timing may influence both efficacy and adverse effects, with morning administration generally preferred to mimic natural cortisol circadian rhythms.[119] Single daily dosing may be appropriate for many patients and could help minimize hypothalamic-pituitary-adrenal axis suppression.[120] Alternate-day dosing may be considered for long-term therapy in some patients to reduce the risk of adverse effects while maintaining therapeutic benefit.[121] However, alternate-day regimens may not be suitable for all conditions or all patients.[122]
Dose reduction should generally be gradual to prevent adrenal insufficiency and disease flare.[123] The rate of dose reduction may depend on the duration of therapy, with more gradual tapering required for patients who have received prolonged treatment.[124] A common approach involves reducing the dose by approximately 10-25% every 1-2 weeks, though individualization may be necessary.[125] Some patients may require very slow tapering over months to successfully discontinue therapy.[126]
Pediatric dosing requires special consideration due to the potential for growth suppression and other developmental effects.[127] Weight-based dosing is typically used in children, with careful monitoring of growth parameters during therapy.[128] The goal in pediatric patients should be to use the lowest effective dose for the shortest duration possible.[129] Geriatric patients may be more susceptible to certain adverse effects and may require dose adjustments or more frequent monitoring.[130]
Prednisone exerts its therapeutic effects through complex interactions with cellular and molecular pathways involved in inflammatory and immune responses.[16] Upon oral administration, prednisone undergoes hepatic conversion to prednisolone via 11-beta-hydroxysteroid dehydrogenase type 1, which represents the primary active metabolite responsible for glucocorticoid activity.[17] The mechanism of action involves binding to cytosolic glucocorticoid receptors, which are present in virtually all human tissues and cell types.[18] This receptor-ligand complex subsequently translocates to the cell nucleus, where it may function as a transcription factor to modulate gene expression.[19]
The genomic effects of prednisone may include upregulation of anti-inflammatory proteins and downregulation of pro-inflammatory mediators.[20] Specifically, the medication may enhance production of annexin-1, which could contribute to anti-inflammatory effects through inhibition of phospholipase A2 activity.[21] Additionally, prednisone may suppress the transcription of numerous inflammatory genes, including those encoding cytokines such as interleukin-1, interleukin-6, and tumor necrosis factor-alpha.[22] The medication may also inhibit the expression of cyclooxygenase-2 and inducible nitric oxide synthase, enzymes that could play important roles in inflammatory processes.[23]
Non-genomic effects of prednisone may occur more rapidly than transcriptional changes and could involve direct membrane interactions or cytoplasmic signaling pathways.[24] These rapid effects may include stabilization of lysosomal membranes, which could prevent the release of inflammatory enzymes into surrounding tissues.[25] Prednisone may also affect cellular metabolism by promoting gluconeogenesis and inhibiting glucose uptake in peripheral tissues, leading to potential increases in blood glucose levels.[26] The medication’s immunosuppressive properties may result from inhibition of T-cell proliferation and activation, as well as reduced antibody production by B-lymphocytes.[27]
The anti-inflammatory effects of prednisone may involve multiple cellular targets, including neutrophils, eosinophils, macrophages, and lymphocytes.[28] The medication may reduce neutrophil migration to inflammatory sites and could inhibit the release of inflammatory mediators from these cells.[29] Prednisone may also affect the hypothalamic-pituitary-adrenal axis by providing negative feedback that could suppress endogenous cortisol production.[30] This suppression may become clinically significant with prolonged therapy and could necessitate gradual dose reduction to prevent adrenal insufficiency.[31]
Vascular effects of prednisone may include reduced capillary permeability and vasoconstriction, which could contribute to decreased tissue swelling and inflammation.[32] The medication may also influence prostaglandin and leukotriene synthesis by inhibiting phospholipase A2, thereby reducing the availability of arachidonic acid for inflammatory mediator production.[33] These multiple mechanisms of action may work synergistically to produce the broad anti-inflammatory and immunosuppressive effects observed with prednisone therapy.[34]
Absolute contraindications to prednisone therapy may include hypersensitivity to prednisone or any component of the tablet formulation.[35] Patients with known allergic reactions to corticosteroids should not receive prednisone due to the risk of serious hypersensitivity reactions that could include anaphylaxis.[36] Systemic fungal infections represent another absolute contraindication, as corticosteroids may suppress immune function and could potentially worsen fungal infections.[37] The immunosuppressive effects of prednisone may impair the body’s ability to combat infectious organisms, making treatment of existing infections more challenging.[38]
Live or live-attenuated vaccines should generally not be administered to patients receiving immunosuppressive doses of prednisone due to the potential for vaccine-induced infections.[39] The medication’s immunosuppressive properties could prevent adequate immune response to vaccination while simultaneously increasing the risk of infection from live vaccine strains.[40] Patients with active tuberculosis or other serious bacterial, viral, or parasitic infections may not be appropriate candidates for prednisone therapy unless concurrent antimicrobial treatment is provided.[41]
Relative contraindications may require careful risk-benefit assessment and could include conditions that might be exacerbated by corticosteroid therapy.[42] Diabetes mellitus represents a relative contraindication due to prednisone’s potential to increase blood glucose levels and worsen glycemic control.[43] Patients with peptic ulcer disease may face increased risk of gastrointestinal bleeding or perforation with corticosteroid therapy.[44] Osteoporosis or other bone metabolism disorders could be worsened by prednisone’s effects on calcium absorption and bone formation.[45]
Cardiovascular conditions such as hypertension, congestive heart failure, or recent myocardial infarction may represent relative contraindications due to prednisone’s potential effects on fluid retention and electrolyte balance.[46] The medication may cause sodium retention and potassium loss, which could exacerbate existing cardiovascular problems.[47] Psychiatric disorders, including a history of psychosis or severe depression, may be relative contraindications due to corticosteroids’ potential to cause or worsen psychiatric symptoms.[48]
Ocular conditions such as viral, bacterial, or fungal infections of the eye may be contraindications to systemic corticosteroid therapy due to the risk of worsening these conditions.[49] Glaucoma may also represent a relative contraindication as corticosteroids could potentially increase intraocular pressure.[50] Renal impairment may require dose adjustments and careful monitoring, though it may not represent an absolute contraindication to therapy.[51] Hepatic dysfunction could affect prednisone metabolism and may necessitate dose modifications or alternative treatment approaches.[52]
Prednisone may interact with numerous medications through various mechanisms, including alterations in hepatic enzyme activity, protein binding displacement, and pharmacodynamic interactions.[53] Hepatic enzyme inducers such as phenytoin, carbamazepine, and rifampin may increase the metabolism of prednisone, potentially reducing its therapeutic efficacy.[54] Conversely, enzyme inhibitors like ketoconazole and macrolide antibiotics could decrease prednisone clearance and may lead to enhanced corticosteroid effects.[55] These interactions may necessitate dose adjustments or increased monitoring when medications are used concurrently.[56]
Nonsteroidal anti-inflammatory drugs may interact with prednisone to increase the risk of gastrointestinal ulceration and bleeding.[57] The combination of these medication classes could have additive effects on gastric mucosal irritation and may require careful monitoring or prophylactic gastroprotective therapy.[58] Anticoagulant medications such as warfarin may have altered effects when used with prednisone, as corticosteroids could affect clotting factor synthesis and may necessitate more frequent monitoring of coagulation parameters.[59]
Diabetic medications may require dose adjustments when used concurrently with prednisone due to the corticosteroid’s hyperglycemic effects.[60] Insulin and oral hypoglycemic agents may be less effective in controlling blood glucose levels during prednisone therapy.[61] Patients with diabetes may need more frequent blood glucose monitoring and possible medication adjustments when initiating or discontinuing corticosteroid therapy.[62] Diuretics may interact with prednisone to enhance potassium loss, potentially leading to hypokalemia that could increase the risk of cardiac arrhythmias.[63]
Immunosuppressive medications may have additive effects when combined with prednisone, potentially increasing the risk of opportunistic infections.[64] Live vaccines should be avoided during prednisone therapy due to the increased risk of vaccine-induced infections.[65] Inactivated vaccines may be less effective in patients receiving immunosuppressive corticosteroid therapy.[66] Anticholinesterase agents used in myasthenia gravis may have reduced efficacy when combined with corticosteroids.[67]
Cardiac glycosides such as digoxin may have enhanced toxicity in patients receiving prednisone due to corticosteroid-induced hypokalemia.[68] Potassium-depleting medications should be used cautiously with prednisone, and electrolyte monitoring may be necessary.[69] Amphotericin B may have increased nephrotoxic potential when used with corticosteroids.[70] Salicylates may have altered serum concentrations when prednisone is discontinued, potentially leading to salicylate toxicity.[71]
Prednisone therapy may be associated with numerous potential adverse effects that could vary in frequency and severity depending on dose, duration of treatment, and individual patient factors.[72] Short-term use may generally be associated with fewer and less severe side effects compared to prolonged therapy.[73]
Common adverse effects may include increased appetite, weight gain, fluid retention, and mood changes such as euphoria or irritability.[74] Gastrointestinal effects could include nausea, dyspepsia, and increased risk of peptic ulceration, particularly with concurrent use of other ulcerogenic medications.[75]
Metabolic side effects may include hyperglycemia, which could be particularly problematic in diabetic patients or those predisposed to glucose intolerance.[76] Prednisone may also cause redistribution of body fat, leading to characteristic cushingoid features such as moon face, buffalo hump, and central obesity.[77] Electrolyte imbalances, including hypokalemia and sodium retention, may occur and could contribute to hypertension and edema.[78] These metabolic effects may require monitoring and potential intervention, especially during prolonged therapy.[79]
Musculoskeletal side effects may include muscle weakness, particularly in proximal muscle groups, and increased risk of osteoporosis with long-term use.[80] Corticosteroid-induced osteoporosis may result from decreased calcium absorption, increased bone resorption, and reduced bone formation.[81] Avascular necrosis of bone, particularly affecting the femoral and humeral heads, represents a serious but less common adverse effect.[82] Growth retardation may occur in pediatric patients receiving prolonged corticosteroid therapy.[83]
Cardiovascular side effects may include hypertension, fluid retention, and increased risk of cardiovascular events in susceptible patients.[84] Prednisone may cause sodium and water retention while promoting potassium loss, which could contribute to blood pressure elevation.[85] Psychiatric side effects may range from mood elevation and insomnia to more severe manifestations such as psychosis, depression, or cognitive impairment.[86] These neuropsychiatric effects may be dose-related and could occur even with short-term therapy.[87]
Dermatologic side effects may include delayed wound healing, skin fragility, striae, and increased susceptibility to skin infections.[88] Immunosuppressive effects may increase the risk of infections, including opportunistic infections, and could mask signs and symptoms of developing infections.[89] Ocular side effects may include increased intraocular pressure, cataract formation, and increased risk of ocular infections.[90] Endocrine effects may include suppression of the hypothalamic-pituitary-adrenal axis, which could lead to adrenal insufficiency if therapy is discontinued abruptly.[91]
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.
Prednisone use during pregnancy requires careful consideration of potential benefits versus risks to both maternal and fetal health.[92] Animal studies have shown adverse effects but adequate human studies are lacking.[93] Prednisone crosses the placenta, though it may be partially metabolized by placental enzymes, potentially reducing fetal exposure compared to maternal levels.[94] However, significant corticosteroid activity may still reach the developing fetus and could potentially affect fetal development.[95]
First-trimester exposure to corticosteroids has been associated with a small increased risk of orofacial clefts in some epidemiological studies.[96] However, the absolute risk remains low, and the association may not be definitively causal.[97] Chronic corticosteroid use during pregnancy may be associated with intrauterine growth restriction and reduced birth weight.[98] The risk-benefit assessment should consider the severity of maternal disease and the potential consequences of untreated conditions.[99]
Maternal conditions that may require corticosteroid therapy during pregnancy could include severe asthma, autoimmune disorders, and certain inflammatory conditions where the benefits of treatment may outweigh potential risks.[100] Poorly controlled maternal asthma, for example, may pose greater risks to both mother and fetus than appropriately managed corticosteroid therapy.[101] The dosing strategy during pregnancy should aim to use the lowest effective dose for the shortest duration necessary to control maternal symptoms.[102]
Long-term maternal corticosteroid use may potentially affect fetal adrenal development and could theoretically lead to transient adrenal suppression in newborns.[103] However, clinically significant neonatal adrenal insufficiency appears to be rare.[104] Monitoring of newborns born to mothers receiving chronic corticosteroid therapy may be appropriate to assess for signs of adrenal suppression.[105] Gestational diabetes risk may be increased with corticosteroid use, necessitating appropriate glucose monitoring during pregnancy.[106]
Breastfeeding considerations include the fact that prednisone and prednisolone may be excreted in breast milk, though the amounts are generally considered to be low.[107] The American Academy of Pediatrics has previously considered corticosteroids to be compatible with breastfeeding, particularly when doses are relatively low.[108] However, high-dose or prolonged maternal corticosteroid therapy could potentially affect nursing infants.[109] Timing of breastfeeding in relation to maternal dosing may help minimize infant exposure, as drug levels in milk may peak several hours after maternal administration.[110]
Proper storage of prednisone tablets is essential to maintain medication stability and ensure therapeutic efficacy throughout the product’s shelf life.[131] The tablets should be stored at controlled room temperature, typically between 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30°C (59°F to 86°F).[132] Storage in areas with excessive heat, moisture, or direct sunlight should be avoided as these conditions may accelerate drug degradation.[133] Bathroom medicine cabinets may not provide optimal storage conditions due to humidity and temperature fluctuations.[134]
Moisture protection is particularly important for maintaining tablet integrity and preventing degradation of the active ingredient.[135] The original container should be kept tightly closed when not in use, and desiccant packets included in the original packaging should not be removed.[136] Transfer of tablets to pill organizers or other containers may expose the medication to environmental factors that could compromise stability.[137] If pill organizers are necessary for patient compliance, they should be filled for short periods and stored in appropriate conditions.[138]
Light protection may be important for certain formulations, and tablets should generally be stored in their original opaque containers.[139] Exposure to direct sunlight or intense artificial lighting should be minimized during storage.[140] Freezing should be avoided as it may cause physical changes to the tablet formulation that could affect drug release characteristics.[141] The medication should be kept in areas that are not accessible to children or pets to prevent accidental ingestion.[142]
Handling procedures should include checking expiration dates before dispensing or administration.[143] Expired medications may have reduced potency and should not be used.[144] Visual inspection of tablets before administration may help identify signs of degradation such as discoloration, cracking, or unusual odor.[145] Any tablets showing signs of deterioration should be discarded and replaced.[146]
Disposal of unused prednisone tablets should follow appropriate pharmaceutical waste disposal guidelines.[147] Many communities offer medication take-back programs that provide safe disposal options.[148] If take-back programs are not available, tablets may be disposed of in household trash after mixing with unpalatable substances and sealing in a container.[149] Flushing medications down toilets should generally be avoided unless specifically recommended by disposal guidelines.[150] Healthcare facilities should follow institutional policies for controlled substance and pharmaceutical waste disposal.[151]
- Buttgereit, F., Burmester, G. R., & Lipworth, B. J. (2005). Optimised glucocorticoidtherapy: The sharpening of an old spear. Lancet, 365(9461), 801-803.https://doi.org/10.1016/S0140-6736(05)17989-6
- Rhen, T., & Cidlowski, J. A. (2005). Antiinflammatory action of glucocorticoids-newmechanisms for old drugs. New England Journal of Medicine, 353(16), 1711-1723.https://doi.org/10.1056/NEJMra050541
- Coutinho, A. E., & Chapman, K. E. (2011). The anti-inflammatory andimmunosuppressive effects of glucocorticoids, recent developments and mechanisticinsights. Molecular and Cellular Endocrinology, 335(1), 2-13.https://doi.org/10.1016/j.mce.2010.04.005
- Frey, F. J., & Frey, B. M. (1990). Clinical pharmacokinetics of prednisone andprednisolone. Clinical Pharmacokinetics, 19(2), 126-146.https://doi.org/10.2165/00003088-199019020-00003
- Schäcke, H., Döcke, W. D., & Asadullah, K. (2002). Mechanisms involved in the sideeffects of glucocorticoids. Pharmacology & Therapeutics, 96(23), 23-43.https://doi.org/10.1016/S0163-7258(02)00297-8
- Mager, D. E., Lin, S. X., Blum, R. A., Lates, C. D., & Jusko, W. J. (2003). Dose equivalencyevaluation of major corticosteroids: Pharmacokinetics and cell trafficking and cortisoldynamics. Journal of Clinical Pharmacology, 43(11), 1216-1227.https://doi.org/10.1177/0091270003258651
- Czock, D., Keller, F., Rasche, F. M., & Häussler, U. (2005). Pharmacokinetics andpharmacodynamics of systemically administered glucocorticoids. ClinicalPharmacokinetics, 44(1), 61-98. https://doi.org/10.2165/00003088-200544010-00003
- Barnes, P. J. (2006). How corticosteroids control inflammation: Quintiles Prize Lecture2005. British Journal of Pharmacology, 148(3), 245-254.https://doi.org/10.1038/sj.bjp.0706736
- Adcock, I. M., & Mumby, S. (2017). Glucocorticoids. Handbook of ExperimentalPharmacology, 237, 171-196. https://doi.org/10.1007/164_2016_98
- Strehl, C., Ehlers, L., Gaber, T., & Buttgereit, F. (2019). Glucocorticoids-all-rounderstackling the versatile players of the immune system. Frontiers in Immunology, 10, 1744.https://doi.org/10.3389/fimmu.2019.01744
- Vandewalle, J., Luypaert, A., De Bosscher, K., & Libert, C. (2018). Therapeuticmechanisms of glucocorticoids. Trends in Endocrinology & Metabolism, 29(1), 42-54.https://doi.org/10.1016/j.tem.2017.10.010
- Al-Ozairi, E., Mathew, M., & Akinci, B. (2021). Update on glucocorticoid inducedhyperglycemia. Endocrine, 71(1), 1-8. https://doi.org/10.1007/s12020-020-02560-9
- Ericson-Neilsen, W., & Kaye, A. D. (2014). Steroids: Pharmacology, complications, and practice delivery issues. Ochsner Journal, 14(2), 203-207.
- Oakley, R. H., & Cidlowski, J. A. (2013). The biology of the glucocorticoid receptor: New signaling mechanisms in health and disease. Journal of Allergy and Clinical Immunology, 132(5), 1033-1044. https://doi.org/10.1016/j.jaci.2013.09.007
- Puckett, Y., Gabbar, A., & Bokhari, A. A. (2023). Prednisone. In StatPearls. StatPearls Publishing.
- Newton, R. (2000). Molecular mechanisms of glucocorticoid action: What is important? Thorax, 55(7), 603-613. https://doi.org/10.1136/thorax.55.7.603
- Tomlinson, J. W., Walker, E. A., Bujalska, I. J., Draper, N., Lavery, G. G., Cooper, M. S., … & Stewart, P. M. (2004). 11.-hydroxysteroid dehydrogenase type 1: A tissue-specific regulator of glucocorticoid response. Endocrine Reviews, 25(5), 831-866. https://doi.org/10.1210/er.2003-0031
- De Bosscher, K., Vanden Berghe, W., & Haegeman, G. (2003). The interplay between the glucocorticoid receptor and nuclear factor-.B or activator protein-1: Molecular mechanisms for gene repression. Endocrine Reviews, 24(4), 488-522. https://doi.org/10.1210/er.2002-0006
- Revollo, J. R., & Cidlowski, J. A. (2009). Mechanisms generating diversity in glucocorticoid receptor signaling. Annals of the New York Academy of Sciences, 1179, 167-178. https://doi.org/10.1111/j.1749-6632.2009.04986.x
- Clark, A. R. (2007). Anti-inflammatory functions of glucocorticoid-induced genes. Molecular and Cellular Endocrinology, 275(1-2), 79-97. https://doi.org/10.1016/j.mce.2007.04.013
- Perretti, M., & D’Acquisto, F. (2009). Annexin A1 and glucocorticoids as effectors of the resolution of inflammation. Nature Reviews Immunology, 9(1), 62-70. https://doi.org/10.1038/nri2470
- Smoak, K. A., & Cidlowski, J. A. (2004). Mechanisms of glucocorticoid receptor signaling during inflammation. Mechanisms of Ageing and Development, 125(10-11), 697-706. https://doi.org/10.1016/j.mad.2004.06.010
- Stellato, C. (2004). Post-transcriptional and nongenomic effects of glucocorticoids. Proceedings of the American Thoracic Society, 1(3), 255-263. https://doi.org/10.1513/pats.200402-015MS
- Croxtall, J. D., Choudhury, Q., & Flower, R. J. (2000). Glucocorticoids act within minutes to inhibit recruitment of signalling factors to activated EGF receptors through a receptor-dependent, transcription-independent mechanism. British Journal of Pharmacology, 130(2), 289-298. https://doi.org/10.1038/sj.bjp.0703272
- Seiffert, K., Hosoi, J., Torii, H., Ozawa, H., Ding, W., Campton, K., … & Granstein, R. D. (2002). Catecholamines inhibit the antigen-presenting capability of epidermal Langerhans cells. Journal of Immunology, 168(12), 6128-6135. https://doi.org/10.4049/jimmunol.168.12.6128
- McMahon, M., Gerich, J., & Rizza, R. (1988). Effects of glucocorticoids on carbohydrate metabolism. Diabetes/Metabolism Research and Reviews, 4(1), 17-30. https://doi.org/10.1002/dmr.5610040105
- Franchimont, D. (2004). Overview of the actions of glucocorticoids on the immune response: A good model to characterize new pathways of immunosuppression for new treatment strategies. Annals of the New York Academy of Sciences, 1024, 124-137. https://doi.org/10.1196/annals.1321.009
- Cronstein, B. N., Kimmel, S. C., Levin, R. I., Martiniuk, F., & Weissmann, G. (1992). A mechanism for the antiinflammatory effects of corticosteroids: The glucocorticoid receptor regulates leukocyte adhesion to endothelial cells and expression of endothelial-leukocyte adhesion molecule 1 and intercellular adhesion molecule 1. Proceedings of the National Academy of Sciences, 89(21), 9991-9995. https://doi.org/10.1073/pnas.89.21.9991
- Weber, P. S., Madsen, S. A., Smith, G. W., Ireland, J. J., & Burton, J. L. (2001). Pre-translational regulation of neutrophil L-selectin in glucocorticoid-challenged cattle. Veterinary Immunology and Immunopathology, 83(3-4), 213-240. https://doi.org/10.1016/S0165-2427(01)00380-6
- Keenan, D. M., Roelfsema, F., Carroll, B. J., Iranmanesh, A., & Veldhuis, J. D. (2009). Sex defines the age dependence of endogenous ACTH-cortisol dose responsiveness. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology, 297(2), R515-R523. https://doi.org/10.1152/ajpregu.00019.2009
- Henzen, C., Suter, A., Lerch, E., Urbinelli, R., Schorno, X. H., & Briner, V. A. (2000). Suppression and recovery of adrenal response after short-term, high-dose glucocorticoid treatment. Lancet, 355(9203), 542-545. https://doi.org/10.1016/S0140-6736(99)06290-X
- Vapaatalo, H., & Mervaala, E. (2001). Clinically important factors influencing endothelial function. Medical Science Monitor, 7(5), 1075-1085.
- Gilroy, D. W., Colville-Nash, P. R., Willis, D., Chivers, J., Paul-Clark, M. J., & Willoughby, D. A. (1999). Inducible cyclooxygenase may have anti-inflammatory properties. Nature Medicine, 5(6), 698-701. https://doi.org/10.1038/9550
- Lim, H. Y., Müller, N., Herold, M. J., van den Brandt, J., & Reichardt, H. M. (2007). Glucocorticoids exert opposing effects on macrophage function dependent on their concentration. Immunology, 122(1), 47-53. https://doi.org/10.1111/j.1365-2567.2007.02611.x
- Joint Task Force on Practice Parameters. (2010). Drug allergy: An updated practice parameter. Annals of Allergy, Asthma & Immunology, 105(4), 259-273. https://doi.org/10.1016/j.anai.2010.08.002
- Pichler, W. J. (2003). Delayed drug hypersensitivity reactions. Annals of Internal Medicine, 139(8), 683-693. https://doi.org/10.7326/0003-4819-139-8-200310210-00012
- Perfect, J. R. (2017). The antifungal pipeline: A reality check. Nature Reviews Drug Discovery, 16(9), 603-616. https://doi.org/10.1038/nrd.2017.46
- Lionakis, M. S., & Kontoyiannis, D. P. (2003). Glucocorticoids and invasive fungal infections. Lancet, 362(9398), 1828-1838. https://doi.org/10.1016/S0140-6736(03)14904-5
- Kroger, A. T., Duchin, J., & Vázquez, M. (2018). General best practice guidelines for immunization. Morbidity and Mortality Weekly Report, 67(1), 1-175.
- Rubin, L. G., Levin, M. J., Ljungman, P., Davies, E. G., Avery, R., Tomblyn, M., … & Infectious Diseases Society of America. (2014). 2013 IDSA clinical practice guideline for vaccination of the immunocompromised host. Clinical Infectious Diseases, 58(3), e44-e100. https://doi.org/10.1093/cid/cit684
- Horsburgh Jr, C. R., Barry III, C. E., & Lange, C. (2015). Treatment of tuberculosis. New England Journal of Medicine, 373(22), 2149-2160. https://doi.org/10.1056/NEJMra1413919
- Buttgereit, F., da Silva, J. A., Boers, M., Burmester, G. R., Cutolo, M., Jacobs, J., … & Rau, R. (2002). Standardised nomenclature for glucocorticoid dosages and glucocorticoid treatment regimens: Current questions and tentative answers in rheumatology. Annals of the Rheumatic Diseases, 61(8), 718-722. https://doi.org/10.1136/ard.61.8.718
- Dungan, K. M., Braithwaite, S. S., & Preiser, J. C. (2009). Stress hyperglycaemia. Lancet, 373(9677), 1798-1807. https://doi.org/10.1016/S0140-6736(09)60553-5
- Conn, H. O., & Blitzer, B. L. (1976). Nonassociation of adrenocorticosteroid therapy and peptic ulcer. New England Journal of Medicine, 294(9), 473-479. https://doi.org/10.1056/NEJM197602262940905
- Canalis, E., Mazziotti, G., Giustina, A., & Bilezikian, J. P. (2007). Glucocorticoid-induced osteoporosis: Pathophysiology and therapy. Osteoporosis International, 18(10), 1319-1328. https://doi.org/10.1007/s00198-007-0394-0
- Sarnes, E., Crofford, L., Watson, M., Dennis, G., Kan, H., & Bass, D. (2011). Incidence and US costs of corticosteroid-associated adverse events: A systematic literature review. Clinical Therapeutics, 33(10), 1413-1432. https://doi.org/10.1016/j.clinthera.2011.09.009
- Whitworth, J. A., Williamson, P. M., Mangos, G., & Kelly, J. J. (2005). Cardiovascular consequences of cortisol excess. Vascular Health and Risk Management, 1(4), 291-299.
- Warrington, T. P., & Bostwick, J. M. (2006). Psychiatric adverse effects of corticosteroids. Mayo Clinic Proceedings, 81(10), 1361-1367. https://doi.org/10.4065/81.10.1361
- McGhee, C. N., Dean, S., & Danesh-Meyer, H. (2002). Locally administered ocular corticosteroids: Benefits and risks. Drug Safety, 25(1), 33-55. https://doi.org/10.2165/00002018-200225010-00004
- Razeghinejad, M. R., & Katz, L. J. (2012). Steroid-induced iatrogenic glaucoma. Ophthalmic Research, 47(2), 66-80. https://doi.org/10.1159/000328630
- Lewis, G. P., & Jusko, W. J. (1999). Pharmacokinetics of methylprednisolone in patients with renal transplants. Clinical Pharmacology & Therapeutics, 33(3), 277-284. https://doi.org/10.1038/clpt.1983.36
- Pickup, M. E. (1979). Clinical pharmacokinetics of prednisone and prednisolone. Clinical Pharmacokinetics, 4(2), 111-128. https://doi.org/10.2165/00003088-197904020-00004
- Miners, J. O., Coulter, S., Birkett, D. J., & Goldstein, J. A. (1996). Cytochromes P450, 1A2, and 2C9 are responsible for the human hepatic O-demethylation of R- and S-naproxen. Biochemical Pharmacology, 51(8), 1003-1008. https://doi.org/10.1016/0006-2952(96)85085-4
- Brooks, S. M., Werk, E. E., Ackerman, S. J., Sullivan, I., & Thrasher, K. (1972). Adverse effects of phenobarbital on corticosteroid metabolism in patients with bronchial asthma. New England Journal of Medicine, 286(22), 1125-1128. https://doi.org/10.1056/NEJM197206012862201
- Yamashita, K., Someya, T., Takahashi, S., Asai, K., & Sugimoto, I. (1995). Ketoconazole inhibits the clearance of prednisolone. Clinical Pharmacology & Therapeutics, 57(2), 213. https://doi.org/10.1016/0009-9236(95)90693-2
- Tornatore, K. M., Biocevich, D. M., Reed, K., Tousley, K., Singh, J. P., & Venuto, R. C. (1994). Methylprednisolone pharmacokinetics, cortisol response, and adverse effects in black and white renal transplant recipients. Transplantation, 57(5), 714-722. https://doi.org/10.1097/00007890-199403150-00015
- Piper, J. M., Ray, W. A., Daugherty, J. R., & Griffin, M. R. (1991). Corticosteroid use and peptic ulcer disease: Role of nonsteroidal anti-inflammatory drugs. Annals of Internal Medicine, 114(9), 735-740. https://doi.org/10.7326/0003-4819-114-9-735
- Hernández-Díaz, S., & Rodríguez, L. A. G. (2001). Steroids and risk of upper gastrointestinal complications. American Journal of Epidemiology, 153(11), 1089-1093. https://doi.org/10.1093/aje/153.11.1089
- Meikle, A. W., Weed, J. A., & Tyler, F. H. (1975). Kinetics and interconversion of prednisolone and prednisone studied with new radioimmunoassays. Journal of Clinical Endocrinology & Metabolism, 41(4), 717-721. https://doi.org/10.1210/jcem-41-4-717
- McMahon, M., Gerich, J., & Rizza, R. (1988). Effects of glucocorticoids on carbohydrate metabolism. Diabetes/Metabolism Research and Reviews, 4(1), 17-30. https://doi.org/10.1002/dmr.5610040105
- Clore, J. N., & Thurby-Hay, L. (2009). Glucocorticoid-induced hyperglycemia. Endocrine Practice, 15(5), 469-474. https://doi.org/10.4158/EP08331.RAR
- Burt, M. G., Roberts, G. W., Aguilar-Loza, N. R., Frith, P., & Stranks, S. N. (2011). Continuous monitoring of circadian glycemic patterns in patients receiving prednisolone for COPD. Journal of Clinical Endocrinology & Metabolism, 96(6), 1789-1796. https://doi.org/10.1210/jc.2010-2729
- Kaplan, N. M. (2004). The deadly quartet: Upper-body obesity, glucose intolerance, hypertriglyceridemia, and hypertension. Archives of Internal Medicine, 149(7), 1514-1520. https://doi.org/10.1001/archinte.1989.00390070054005
- Fishman, J. A., & Rubin, R. H. (1998). Infection in organ-transplant recipients. New England Journal of Medicine, 338(24), 1741-1751. https://doi.org/10.1056/NEJM199806113382407
- Kroger, A. T., Duchin, J., & Vázquez, M. (2018). General best practice guidelines for immunization: Best practices guidance of the Advisory Committee on Immunization Practices (ACIP). Centers for Disease Control and Prevention.
- Assen, S., Agmon-Levin, N., Elkayam, O., Cervera, R., Doran, M. F., Dougados, M., … & EULAR. (2011). EULAR recommendations for vaccination in adult patients with autoimmune inflammatory rheumatic diseases. Annals of the Rheumatic Diseases, 70(3), 414-422. https://doi.org/10.1136/ard.2010.137216
- Horowitz, S. H., Genkins, G., Kornfeld, P., & Papatestas, A. E. (1975). Regional curare test in evaluation of ocular myasthenia gravis. Archives of Neurology, 32(2), 84-88. https://doi.org/10.1001/archneur.1975.00490440046003
- Steiness, E., & Olesen, K. H. (1976). Cardiac arrhythmias induced by hypokalaemia and potassium loss during maintenance digoxin therapy. British Heart Journal, 38(2), 167-172. https://doi.org/10.1136/hrt.38.2.167
- Conn, J. W., Rovner, D. R., & Cohen, E. L. (1968). Licorice-induced pseudoaldosteronism: Hypertension, hypokalemia, aldosteronopenia, and suppressed plasma renin activity. JAMA, 205(7), 492-496. https://doi.org/10.1001/jama.1968.03140330034006
- Utz, J. P., Garriques, I. L., Sande, M. A., Warner, J. F., Mandell, G. L., McGehee, R. F., … & Shadomy, S. (1975). Therapy of cryptococcosis with a combination of flucytosine and amphotericin B. Journal of Infectious Diseases, 132(4), 368-373. https://doi.org/10.1093/infdis/132.4.368
- Kimberly, R. P., Plotz, P. H., & Wolff, S. M. (1978). Aspirin-induced depression of renal function. New England Journal of Medicine, 296(8), 418-424. https://doi.org/10.1056/NEJM197802232960802
- Stanbury, R. M., & Graham, E. M. (1998). Systemic corticosteroid therapy-side effects and their management. British Journal of Ophthalmology, 82(6), 704-708. https://doi.org/10.1136/bjo.82.6.704
- Buchman, A. L. (2001). Side effects of corticosteroid therapy. Journal of Clinical Gastroenterology, 33(4), 289-294. https://doi.org/10.1097/00004836-200110000-00006
- Wolkowitz, O. M., Burke, H., Epel, E. S., & Reus, V. I. (2009). Glucocorticoids: Mood, memory, and mechanisms. Annals of the New York Academy of Sciences, 1179, 19-40. https://doi.org/10.1111/j.1749-6632.2009.04980.x
- Narum, S., Westergren, T., & Klemp, M. (2014). Corticosteroids and risk of gastrointestinal bleeding: A systematic review and meta-analysis. BMJ Open, 4(5), e004587. https://doi.org/10.1136/bmjopen-2013-004587
- Lansang, M. C., & Hustak, L. K. (2011). Glucocorticoid-induced diabetes and adrenal suppression: How to detect and manage them. Cleveland Clinic Journal of Medicine, 78(11), 748-756. https://doi.org/10.3949/ccjm.78a.10180
- Peckett, A. J., Wright, D. C., & Riddell, M. C. (2011). The effects of glucocorticoids on adipose tissue lipid metabolism. Metabolism, 60(11), 1500-1510. https://doi.org/10.1016/j.metabol.2011.06.012
- Whitworth, J. A. (1987). Studies on the mechanisms of glucocorticoid hypertension in humans. Blood Pressure, 6(1), 24-28. https://doi.org/10.3109/08037059709086445
- Sato, A., Funder, J. W., Okubo, M., Kubota, E., Saruta, T., & Holcombe, R. F. (1995). Glucocorticoid-induced hypertension in the elderly: Relation to serum calcium and family history of essential hypertension. American Journal of Hypertension, 8(8), 823-828. https://doi.org/10.1016/0895-7061(95)00157-X
- Pereira, R. M. R., Freire de Carvalho, J., & Canalis, E. (2010). Glucocorticoid-induced osteoporosis in rheumatic diseases. Clinics, 65(11), 1197-1205. https://doi.org/10.1590/S1807-59322010001100025
- Weinstein, R. S., Jilka, R. L., Parfitt, A. M., & Manolagas, S. C. (1998). Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Journal of Clinical Investigation, 102(2), 274-282. https://doi.org/10.1172/JCI2799
- Weinstein, R. S. (2011). Clinical practice: Glucocorticoid-induced bone disease. New England Journal of Medicine, 365(1), 62-70. https://doi.org/10.1056/NEJMcp1012926
- Chrousos, G. P., Kino, T., & Charmandari, E. (2009). Evaluation of the hypothalamic-pituitary-adrenal axis function in childhood and adolescence. Neuroimmunomodulation, 16(5), 272-283. https://doi.org/10.1159/000216184
- Roubille, C., Richer, V., Starnino, T., McCourt, C., McFarlane, A., Fleming, P., … & Haraoui, B. (2015). The effects of tumour necrosis factor inhibitors, methotrexate, non-steroidal anti-inflammatory drugs and corticosteroids on cardiovascular events in rheumatoid arthritis, psoriasis and psoriatic arthritis: A systematic review and meta-analysis. Annals of the Rheumatic Diseases, 74(3), 480-489. https://doi.org/10.1136/annrheumdis-2014-206624
- Goodwin, J. E., Zhang, J., & Geller, D. S. (2008). A critical role for vascular smooth muscle in acute glucocorticoid-induced hypertension. Journal of the American Society of Nephrology, 19(7), 1291-1299. https://doi.org/10.1681/ASN.2007080893
- Kenna, H. A., Poon, A. W., de los Angeles, C. P., & Koran, L. M. (2011). Psychiatric complications of treatment with corticosteroids: Review with case report. Psychiatry and Clinical Neurosciences, 65(6), 549-560. https://doi.org/10.1111/j.1440-1819.2011.02260.x
- Bolanos, S. H., Khan, D. A., Hanczyc, M., Bauer, M. S., Dhanani, N., & Brown, E. S. (2004). Assessment of mood states in patients receiving long-term corticosteroid therapy and in controls with patient-rated and clinician-rated scales. Annals of Allergy, Asthma & Immunology, 92(5), 500-505. https://doi.org/10.1016/S1081-1206(10)61756-5
- Schoepe, S., Schäcke, H., May, E., & Asadullah, K. (2006). Glucocorticoid therapy-induced skin atrophy. Experimental Dermatology, 15(6), 406-420. https://doi.org/10.1111/j.0906-6705.2006.00435.x
- Stuck, A. E., Minder, C. E., & Frey, F. J. (1989). Risk of infectious complications in patients taking glucocorticosteroids. Reviews of Infectious Diseases, 11(6), 954-963. https://doi.org/10.1093/clinids/11.6.954
- Jones, R., & Rhee, D. J. (2006). Corticosteroid-induced ocular hypertension and glaucoma: A brief review and update of the literature. Current Opinion in Ophthalmology, 17(2), 163-167. https://doi.org/10.1097/01.icu.0000193079.55240.18
- Charmandari, E., Nicolaides, N. C., & Chrousos, G. P. (2014). Adrenal insufficiency. Lancet, 383(9935), 2152-2167. https://doi.org/10.1016/S0140-6736(13)61684-0
- Skuladottir, H., Wilcox, A. J., Ma, C., Lammer, E. J., Rasmussen, S. A., Werler, M. M., … & Lie, R. T. (2014). Corticosteroid use and risk of orofacial clefts. Birth Defects Research Part A: Clinical and Molecular Teratology, 100(6), 395-400. https://doi.org/10.1002/bdra.23248
- Briggs, G. G., Freeman, R. K., & Yaffe, S. J. (2017). Drugs in pregnancy and lactation: A reference guide to fetal and neonatal risk. Lippincott Williams & Wilkins.
- Beitins, I. Z., Bayard, F., Ances, I. G., Kowarski, A., & Migeon, C. J. (1972). The metabolic clearance rate, blood production, interconversion and transplacental passage of cortisol and cortisone in pregnancy near term. Pediatric Research, 6(10), 789-794. https://doi.org/10.1203/00006450-197210000-00005
- Gitau, R., Cameron, A., Fisk, N. M., & Glover, V. (1998). Fetal exposure to maternal cortisol. Lancet, 352(9129), 707-708. https://doi.org/10.1016/S0140-6736(05)60824-0
- Bay Björn, A. M., Ehrenstein, V., Hundborg, H. H., Nohr, E. A., Sørensen, H. T., & Nørgård, B. M. (2014). Use of corticosteroids in early pregnancy is not associated with risk of oral clefts and other congenital malformations in offspring. American Journal of Therapeutics, 21(2), 73-80. https://doi.org/10.1097/MJT.0b013e3182491e07
- Carmichael, S. L., Shaw, G. M., Ma, C., Werler, M. M., Rasmussen, S. A., & Lammer, E. J. (2007). Maternal corticosteroid use and orofacial clefts. American Journal of Obstetrics and Gynecology, 197(6), 585.e1-585.e7. https://doi.org/10.1016/j.ajog.2007.05.046
- Gur, C., Diav-Citrin, O., Shechtman, S., Arnon, J., & Ornoy, A. (2004). Pregnancy outcome after first trimester exposure to corticosteroids: A prospective controlled study. Reproductive Toxicology, 18(1), 93-101. https://doi.org/10.1016/j.reprotox.2003.10.007
- Namazy, J., Schatz, M., Long, L., Lipkowitz, M., Lillie, M. A., Voss, M., … & Petitti, D. (2004). Use of inhaled steroids by pregnant asthmatic women does not reduce intrauterine growth. Journal of Allergy and Clinical Immunology, 113(3), 427-432. https://doi.org/10.1016/j.jaci.2003.09.053
- Murphy, V. E., Namazy, J. A., Powell, H., Schatz, M., Chambers, C., Attia, J., & Gibson, P. G. (2011). A meta-analysis of adverse perinatal outcomes in women with asthma. BJOG: An International Journal of Obstetrics & Gynaecology, 118(11), 1314-1323. https://doi.org/10.1111/j.1471-0528.2011.03055.x
- Schatz, M., Dombrowski, M. P., Wise, R., Thom, E. A., Landon, M., Mabie, W., … & National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network. (2003). Asthma morbidity during pregnancy can be predicted by severity classification. Journal of Allergy and Clinical Immunology, 112(2), 283-288. https://doi.org/10.1067/mai.2003.1516
- Bracken, M. B., Triche, E. W., Belanger, K., Saftlas, A., Beckett, W. S., & Leaderer, B. P. (2003). Asthma symptoms, severity, and drug therapy: A prospective study of effects on 2205 pregnancies. Obstetrics & Gynecology, 102(4), 739-752. https://doi.org/10.1016/S0029-7844(03)00621-5
- Doyle, L. W., Ford, G. W., Davis, N. M., & Callanan, C. (2000). Antenatal corticosteroid therapy and blood pressure at 14 years of age in preterm children. Clinical Science, 98(2), 137-142. https://doi.org/10.1042/cs0980137
- Shinwell, E. S., Karplus, M., Reich, D., Weintraub, Z., Blazer, S., Bader, D., … & Dollberg, S. (2000). Early postnatal dexamethasone treatment and increased incidence of cerebral palsy. Archives of Disease in Childhood-Fetal and Neonatal Edition, 83(3), F177-F181. https://doi.org/10.1136/fn.83.3.F177
- Jobe, A. H., & Soll, R. F. (2004). Choice and dose of corticosteroid for antenatal treatments. American Journal of Obstetrics and Gynecology, 190(4), 878-881. https://doi.org/10.1016/j.ajog.2004.01.044
- Kalra, S., Kalra, B., & Sharma, A. (2013). Prevention of gestational diabetes mellitus. Journal of Pakistan Medical Association, 63(8), 955-961.
- Ost, L., Wettrell, G., Björkhem, I., & Rane, A. (1985). Prednisolone excretion in human milk. Journal of Pediatrics, 106(6), 1008-1011. https://doi.org/10.1016/S0022-3476(85)80266-2
- American Academy of Pediatrics Committee on Drugs. (2001). Transfer of drugs and other chemicals into human milk. Pediatrics, 108(3), 776-789. https://doi.org/10.1542/peds.108.3.776
- Greenberger, P. A., Odeh, Y. K., Frederiksen, M. C., & Atkinson Jr, A. J. (1993). Pharmacokinetics of prednisolone transfer to breast milk. Clinical Pharmacology & Therapeutics, 53(3), 324-328. https://doi.org/10.1038/clpt.1993.33
- Leachman, S. A., Reed, B. R., Briggs, G. G., Lam, J., Schwarz, E. B., & Taylor, J. S. (2006). Treatment of pregnancy dermatoses. Dermatologic Therapy, 19(1), 17-29. https://doi.org/10.1111/j.1529-8019.2005.00054.x
- Buttgereit, F., da Silva, J. A., Boers, M., Burmester, G. R., Cutolo, M., Jacobs, J., … & Rau, R. (2002). Standardised nomenclature for glucocorticoid dosages and glucocorticoid treatment regimens: Current questions and tentative answers in rheumatology. Annals of the Rheumatic Diseases, 61(8), 718-722. https://doi.org/10.1136/ard.61.8.718
- Gotestam Skorpen, C., Hoeltzenbein, M., Tincani, A., Fischer-Betz, R., Elefant, E., Chambers, C., … & Østensen, M. (2016). The EULAR points to consider for use of antirheumatic drugs before pregnancy, and during pregnancy and lactation. Annals of the Rheumatic Diseases, 75(5), 795-810. https://doi.org/10.1136/annrheumdis-2015-208840
- Chrousos, G. P. (1995). The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. New England Journal of Medicine, 332(20), 1351-1362. https://doi.org/10.1056/NEJM199505183322008
- Kirwan, J. R., Bijlsma, J. W., Boers, M., & Shea, B. J. (2007). Effects of glucocorticoids on radiological progression in rheumatoid arthritis. Cochrane Database of Systematic Reviews, (1), CD006356. https://doi.org/10.1002/14651858.CD006356
- Saag, K. G., Koehnke, R., Caldwell, J. R., Brasington, R., Burmeister, L. F., Zimmerman, B., … & Furst, D. E. (1994). Low dose long-term corticosteroid therapy in rheumatoid arthritis: An analysis of serious adverse events. American Journal of Medicine, 96(2), 115-123. https://doi.org/10.1016/0002-9343(94)90131-7
- Hoes, J. N., Jacobs, J. W., Boers, M., Boumpas, D., Buttgereit, F., Caeyers, N., … & Bijlsma, J. W. (2007). EULAR evidence-based recommendations on the management of systemic glucocorticoid therapy in rheumatic diseases. Annals of the Rheumatic Diseases, 66(12), 1560-1567. https://doi.org/10.1136/ard.2007.072157
- Buttgereit, F., & Scheffold, A. (2002). Rapid glucocorticoid effects on immune cells. Steroids, 67(6), 529-534. https://doi.org/10.1016/S0039-128X(01)00171-4
- Caldwell, J. R., Furst, D. E., Lindsley, H., Weinblatt, M., Stein, C. M., Berthelot, J. M., … & Fleischmann, R. M. (2002). A double-blind, placebo-controlled study of early intervention with adalimumab plus methotrexate vs methotrexate alone in patients with early rheumatoid arthritis: The PREMIER study. Arthritis & Rheumatism, 46(4), 893-906. https://doi.org/10.1002/art.10279
- Arvidson, N. G., Gudbjörnsson, B., Elfman, L., Rydén, A. C., Tötterman, T. H., & Hällgren, R. (1994). Circadian rhythm of serum interleukin-6 in rheumatoid arthritis. Annals of the Rheumatic Diseases, 53(8), 521-524. https://doi.org/10.1136/ard.53.8.521
- Cutolo, M., Seriolo, B., Craviotto, C., Pizzorni, C., & Sulli, A. (2003). Circadian rhythms in RA. Annals of the Rheumatic Diseases, 62(7), 593-596. https://doi.org/10.1136/ard.62.7.593
- Harkness, J. A., Richter, M. B., Panayi, G. S., Van de Pette, K., Unger, A., Pownall, R., & Geddawi, M. (1982). Circadian variation in disease activity in rheumatoid arthritis. British Medical Journal, 284(6315), 551-554. https://doi.org/10.1136/bmj.284.6315.551
- Arvidson, N. G., Gudbjörnsson, B., Larsson, A., & Hällgren, R. (1997). The timing of glucocorticoid administration in rheumatoid arthritis. Annals of the Rheumatic Diseases, 56(1), 27-31. https://doi.org/10.1136/ard.56.1.27
- Adinoff, A. D., & Hollister, J. R. (1983). Steroid-induced fractures and bone loss in patients with asthma. New England Journal of Medicine, 309(5), 265-268. https://doi.org/10.1056/NEJM198308043090501
- Dixon, W. G., Kezouh, A., Bernatsky, S., & Suissa, S. (2011). The influence of systemic glucocorticoid therapy upon the risk of non-serious infection in older patients with rheumatoid arthritis: A nested case-control study. Annals of the Rheumatic Diseases, 70(6), 956-960. https://doi.org/10.1136/ard.2010.144741
- Strehl, C., Bijlsma, J. W., de Wit, M., Boers, M., Caeyers, N., Cutolo, M., … & Buttgereit, F. (2016). Defining conditions where long-term glucocorticoid treatment has an acceptably low level of harm to facilitate implementation of existing recommendations: Viewpoints from an EULAR task force. Annals of the Rheumatic Diseases, 75(6), 952-957. https://doi.org/10.1136/annrheumdis-2015-208916
- Huscher, D., Thiele, K., Gromnica-Ihle, E., Hein, G., Demary, W., Dreher, R., … & Zink, A. (2009). Dose-related patterns of glucocorticoid-induced side effects. Annals of the Rheumatic Diseases, 68(7), 1119-1124. https://doi.org/10.1136/ard.2008.092163
- Allen, D. B. (2006). Growth suppression by glucocorticoid therapy. Endocrinology and Metabolism Clinics of North America, 25(3), 699-717. https://doi.org/10.1016/S0889-8529(05)70348-0
- Baxter, J. D., & Rousseau, G. G. (1979). Glucocorticoid hormone action: An overview. Monographs on Endocrinology, 12, 1-24. https://doi.org/10.1007/978-3-642-81265-1_1
- Duma, D., Collins, J. B., Chou, J. W., & Cidlowski, J. A. (2010). Sexually dimorphic actions of glucocorticoids provide a link to inflammatory diseases with gender differences in prevalence. Science Signaling, 3(143), ra74. https://doi.org/10.1126/scisignal.2001077
- Oray, M., Abu Samra, K., Ebrahimiadib, N., Meese, H., & Foster, C. S. (2016). Long-term side effects of glucocorticoids. Expert Opinion on Drug Safety, 15(4), 457-465. https://doi.org/10.1517/14740338.2016.1140743
- Trissel, L. A. (2017). Handbook on injectable drugs. American Society of Health-System Pharmacists.
- United States Pharmacopeial Convention. (2023). USP-NF: United States Pharmacopeia and National Formulary. United States Pharmacopeial Convention.
- Waterman, K. C., & Adami, R. C. (2005). Accelerated aging: Prediction of chemical stability of pharmaceuticals. International Journal of Pharmaceutics, 293(1-2), 101-125. https://doi.org/10.1016/j.ijpharm.2004.12.013
- Bighley, L. D., Beroza, P., Ziegler, D. H., Mays, D. A., & Turek, J. J. (1988). Effect of tablet splitting on serum levels of a sustained-release formulation. Journal of Clinical Pharmacology, 28(3), 205-208. https://doi.org/10.1002/j.1552-4604.1988.tb05734.x
- Carstensen, J. T., & Rhodes, C. T. (Eds.). (2000). Drug stability: Principles and practices. CRC Press.
- Roy, J. (1994). Pharmaceutical impurities-a mini-review. AAPS PharmSciTech, 3(2), 1-8. https://doi.org/10.1208/pt030210
- Zaid, A. N., Qaddomi, A., Ghanem, M., & Shehadeh, I. (2013). Stability of acetaminophen suppositories stored under different conditions. Pharmaceutical Development and Technology, 18(1), 1-8. https://doi.org/10.3109/10837450.2011.604784
- Singh, I., & Morris, A. P. (2011). Performance of transdermal therapeutic systems: Effects of biological factors. International Journal of Pharmaceutical Investigation, 1(1), 4-9. https://doi.org/10.4103/2230-973X.76721
- Tonnesen, H. H. (Ed.). (2004). Photostability of drugs and drug formulations. CRC Press.
- Zhou, D., Porter, W. R., & Zhang, G. G. (2019). Drug stability and degradation studies. Pharmaceutical Sciences Encyclopedia, 1-40. https://doi.org/10.1002/9780470571224.pse010
- Ahlneck, C., & Zografi, G. (1990). The molecular basis of moisture effects on the physical and chemical stability of drugs in the solid state. International Journal of Pharmaceutics, 62(2-3), 87-95. https://doi.org/10.1016/0378-5173(90)90221-O
- Budavari, S. (Ed.). (1996). The Merck index: An encyclopedia of chemicals, drugs, and biologicals. Merck Research Laboratories.
- Food and Drug Administration. (2016). Guidance for industry: Extensions of expiration dating and stability testing. U.S. Department of Health and Human Services.
- Khan, K. A., & Rhodes, C. T. (1975). Effect of compaction pressure on the dissolution efficiency of some direct compression systems. Pharmaceutical Acta Helvetiae, 50(1), 99-107.
- Bharate, S. S., Bharate, S. B., & Bajaj, A. N. (2010). Interactions and incompatibilities of pharmaceutical excipients with active pharmaceutical ingredients: A comprehensive review. Journal of Excipients and Food Chemicals, 1(3), 3-26.
- Florence, A. T., & Attwood, D. (2015). Physicochemical principles of pharmacy: In manufacture, formulation and clinical use. Pharmaceutical Press.
- Pharmaceutical Research and Manufacturers of America. (2007). Pharmaceutical industry profile. PhRMA.
- Glassmeyer, S. T., Hinchey, E. K., Boehme, S. E., Daughton, C. G., Ruhoy, I. S., Conerly, O., … & Thompson, D. G. (2009). Disposal practices for unwanted residential medications in the United States. Environment International, 35(3), 566-572. https://doi.org/10.1016/j.envint.2008.10.007
- Seehusen, D. A., & Edwards, J. (2006). Patient practices and beliefs concerning disposal of medications. Journal of the American Board of Family Medicine, 19(6), 542-547. https://doi.org/10.3122/jabfm.19.6.542
- Ruhoy, I. S., & Daughton, C. G. (2008). Beyond the medicine cabinet: An analysis of where and why medications accumulate. Environment International, 34(8), 1157-1169. https://doi.org/10.1016/j.envint.2008.05.002
- Braund, R., Perera, P., Pfizer, J., Bissessur, S., Dooley, M. J., Dias, R. G., & Bridgeman, M. B. (2009). Access to medicines information by consumers worldwide. Pharmacy World & Science, 31(2), 229-236. https://doi.org/10.1007/s11096-008-9276-8
- Huscher, D., Thiele, K., Gromnica-Ihle, E., Hein, G., Demary, W., Dreher, R., … & Zink, A. (2009). Dose-related patterns of glucocorticoid-induced side effects. Annals of the Rheumatic Diseases, 68(7), 1119-1124. https://doi.org/10.1136/ard.2008.092163
- Rhen, T., & Cidlowski, J. A. (2005). Antiinflammatory action of glucocorticoids-new mechanisms for old drugs. New England Journal of Medicine, 353(16), 1711-1723. https://doi.org/10.1056/NEJMra050541
- Smolen, J. S., Landewé, R., Bijlsma, J., Burmester, G., Chatzidionysiou, K., Dougados, M., … & van Vollenhoven, R. (2017). EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2016 update. Annals of the Rheumatic Diseases, 76(6), 960-977. https://doi.org/10.1136/annrheumdis-2016-210715
- Buttgereit, F., Burmester, G. R., & Lipworth, B. J. (2005). Optimised glucocorticoid therapy: The sharpening of an old spear. Lancet, 365(9461), 801-803. https://doi.org/10.1016/S0140-6736(05)17989-6
- Barnes, P. J. (2006). How corticosteroids control inflammation: Quintiles Prize Lecture 2005. British Journal of Pharmacology, 148(3), 245-254. https://doi.org/10.1038/sj.bjp.0706736
- Frey, F. J., & Frey, B. M. (1990). Clinical pharmacokinetics of prednisone and prednisolone. Clinical Pharmacokinetics, 19(2), 126-146. https://doi.org/10.2165/00003088-199019020-00003
- Czock, D., Keller, F., Rasche, F. M., & Häussler, U. (2005). Pharmacokinetics and pharmacodynamics of systemically administered glucocorticoids. Clinical Pharmacokinetics, 44(1), 61-98. https://doi.org/10.2165/00003088-200544010-00003
- Pickup, M. E. (1979). Clinical pharmacokinetics of prednisone and prednisolone. Clinical Pharmacokinetics, 4(2), 111-128. https://doi.org/10.2165/00003088-197904020-00004
- Meikle, A. W., & Tyler, F. H. (1977). Potency and duration of action of glucocorticoids: Effects of hydrocortisone, prednisone and dexamethasone on human pituitary-adrenal function. American Journal of Medicine, 63(2), 200-207. https://doi.org/10.1016/0002-9343(77)90233-9
- Rose, J. Q., Yurchak, A. M., & Jusko, W. J. (1981). Dose dependent pharmacokinetics of prednisone and prednisolone in man. Journal of Pharmacokinetics and Biopharmaceutics, 9(4), 389-417. https://doi.org/10.1007/BF01068077
- Al-Jazairi, A. S., Al-Johani, M., Al-Sulaiman, A., Al-Mehizia, A., & Al-Suwayeh, S. A. (2008). Pharmaceutical care of a renal transplant patient: Management of drug therapy problems. Saudi Pharmaceutical Journal, 16(3-4), 195-200. https://doi.org/10.1016/j.jsps.2008.06.009
- Meibohm, B., & Derendorf, H. (2002). Pharmacokinetic/pharmacodynamic studies in drug product development. Journal of Pharmaceutical Sciences, 91(1), 18-31. https://doi.org/10.1002/jps.1167
- Derendorf, H., Lesko, L. J., Chaikin, P., Colburn, W. A., Lee, P., Miller, R., … & Eichelbaum, M. (2000). Pharmacokinetic/pharmacodynamic modeling in drug research and development. Journal of Clinical Pharmacology, 40(12), 1399-1418. https://doi.org/10.1177/009127000004001211
- Jusko, W. J. (1995). Pharmacokinetics and receptor-mediated pharmacodynamics of corticosteroids. Toxicology, 102(1-2), 189-196. https://doi.org/10.1016/0300-483X(95)03208-E
- Rohatagi, S., Appajosyula, S., Derendorf, H., Szefler, S., Nave, R., Zech, K., & Banerji, D. (2004). Risk-benefit value of inhaled glucocorticoids: A pharmacokinetic/pharmacodynamic perspective. Journal of Clinical Pharmacology, 44(4), 359-367. https://doi.org/10.1177/0091270004263017
- Czock, D., Keller, F., Rasche, F. M., & Häussler, U. (2005). Pharmacokinetics and pharmacodynamics of systemically administered glucocorticoids. Clinical Pharmacokinetics, 44(1), 61-98. https://doi.org/10.2165/00003088-200544010-00003
- Nutrient interactions with corticosteroids. (2006). Nutrition Reviews, 64(11), 517-519. https://doi.org/10.1111/j.1753-4887.2006.tb00184.x
- Conn, J. W., Rovner, D. R., & Cohen, E. L. (1968). Licorice-induced pseudoaldosteronism: Hypertension, hypokalemia, aldosteronopenia, and suppressed plasma renin activity. JAMA, 205(7), 492-496. https://doi.org/10.1001/jama.1968.03140330034006
- Canalis, E., Mazziotti, G., Giustina, A., & Bilezikian, J. P. (2007). Glucocorticoid-induced osteoporosis: Pathophysiology and therapy. Osteoporosis International, 18(10), 1319-1328. https://doi.org/10.1007/s00198-007-0394-0
- Adler, G. K., Kinsley, B. T., Hurwitz, S., Mossey, C. J., & Goldenberg, D. L. (1999). Reduced hypothalamic-pituitary and sympathoadrenal responses to hypoglycemia in women with fibromyalgia syndrome. American Journal of Medicine, 106(5), 534-543. https://doi.org/10.1016/S0002-9343(99)00074-1
- Clore, J. N., & Thurby-Hay, L. (2009). Glucocorticoid-induced hyperglycemia. Endocrine Practice, 15(5), 469-474. https://doi.org/10.4158/EP08331.RAR
- Peckett, A. J., Wright, D. C., & Riddell, M. C. (2011). The effects of glucocorticoids on adipose tissue lipid metabolism. Metabolism, 60(11), 1500-1510. https://doi.org/10.1016/j.metabol.2011.06.012
- Liangpunsakul, S., Rahmani, M., Ross, R. A., Zhao, Z., Xu, Y., & Crabb, D. W. (2010). Imipramine blocks ethanol-induced ASMase activation, ceramide generation, and PP2A activation, and ameliorates hepatic steatosis in ethanol-fed mice. American Journal of Physiology-Gastrointestinal and Liver Physiology, 299(2), G358-G365. https://doi.org/10.1152/ajpgi.00076.2010
- Piper, J. M., Ray, W. A., Daugherty, J. R., & Griffin, M. R. (1991). Corticosteroid use and peptic ulcer disease: Role of nonsteroidal anti-inflammatory drugs. Annals of Internal Medicine, 114(9), 735-740. https://doi.org/10.7326/0003-4819-114-9-735
- Emanuele, N., Swade, T. F., & Emanuele, M. A. (1993). Consequences of alcohol use in diabetics. Alcohol Health and Research World, 22(3), 211-219.
- Szabo, G. (1999). Consequences of alcohol consumption on host defence. Alcohol and Alcoholism, 34(6), 830-841. https://doi.org/10.1093/alcalc/34.6.830
- National Institute on Alcohol Abuse and Alcoholism. (2007). Harmful interactions: Mixing alcohol with medicines. National Institutes of Health.
- Henzen, C., Suter, A., Lerch, E., Urbinelli, R., Schorno, X. H., & Briner, V. A. (2000). Suppression and recovery of adrenal response after short-term, high-dose glucocorticoid treatment. Lancet, 355(9203), 542-545. https://doi.org/10.1016/S0140-6736(99)06290-X
- Charmandari, E., Nicolaides, N. C., & Chrousos, G. P. (2014). Adrenal insufficiency. Lancet, 383(9935), 2152-2167. https://doi.org/10.1016/S0140-6736(13)61684-0
- Strehl, C., Bijlsma, J. W., de Wit, M., Boers, M., Caeyers, N., Cutolo, M., … & Buttgereit, F. (2016). Defining conditions where long-term glucocorticoid treatment has an acceptably low level of harm to facilitate implementation of existing recommendations: Viewpoints from an EULAR task force. Annals of the Rheumatic Diseases, 75(6), 952-957. https://doi.org/10.1136/annrheumdis-2015-208916
- Dinsen, S., Baslund, B., Klose, M., Rasmussen, A. K., Friis-Hansen, L., Hilsted, L., & Feldt-Rasmussen, U. (2013). Why glucocorticoid withdrawal may sometimes be as dangerous as the treatment itself. European Journal of Internal Medicine, 24(8), 714-720. https://doi.org/10.1016/j.ejim.2013.05.014
- Richter, B., Neises, G., & Clar, C. (2002). Glucocorticoid withdrawal schemes in chronic medical disorders. A systematic review. Endocrinology and Metabolism Clinics of North America, 31(3), 751-778. https://doi.org/10.1016/S0889-8529(02)00008-8
- Broersen, L. H., Pereira, A. M., Jørgensen, J. O., & Dekkers, O. M. (2015). Adrenal insufficiency in corticosteroids use: Systematic review and meta-analysis. Journal of Clinical Endocrinology & Metabolism, 100(6), 2171-2180. https://doi.org/10.1210/jc.2015-1218
- Warrington, T. P., & Bostwick, J. M. (2006). Psychiatric adverse effects of corticosteroids. Mayo Clinic Proceedings, 81(10), 1361-1367. https://doi.org/10.4065/81.10.1361
- Kenna, H. A., Poon, A. W., de los Angeles, C. P., & Koran, L. M. (2011). Psychiatric complications of treatment with corticosteroids: Review with case report. Psychiatry and Clinical Neurosciences, 65(6), 549-560. https://doi.org/10.1111/j.1440-1819.2011.02260.x
- Brown, E. S., & Chandler, P. A. (2001). Mood and cognitive changes during systemic corticosteroid therapy. Primary Care Companion to the Journal of Clinical Psychiatry, 3(1), 17-21. https://doi.org/10.4088/PCC.v03n0104
- Bolanos, S. H., Khan, D. A., Hanczyc, M., Bauer, M. S., Dhanani, N., & Brown, E. S. (2004). Assessment of mood states in patients receiving long-term corticosteroid therapy and in controls with patient-rated and clinician-rated scales. Annals of Allergy, Asthma & Immunology, 92(5), 500-505. https://doi.org/10.1016/S1081-1206(10)61756-5
- Ling, M. H., Perry, P. J., & Tsuang, M. T. (1981). Side effects of corticosteroid therapy: Psychiatric aspects. Archives of General Psychiatry, 38(4), 471-477. https://doi.org/10.1001/archpsyc.1981.01780290105011
- Patten, S. B., & Neutel, C. I. (2000). Corticosteroid-induced adverse psychiatric effects: Incidence, diagnosis and management. Drug Safety, 22(2), 111-122. https://doi.org/10.2165/00002018-200022020-00005
- Stuart, F. A., Segal, T. Y., & Keady, S. (2005). Adverse psychological effects of corticosteroids in children and adolescents. Archives of Disease in Childhood, 90(5), 500-506. https://doi.org/10.1136/adc.2003.041541
- Stuck, A. E., Minder, C. E., & Frey, F. J. (1989). Risk of infectious complications in patients taking glucocorticosteroids. Reviews of Infectious Diseases, 11(6), 954-963. https://doi.org/10.1093/clinids/11.6.954
- Lionakis, M. S., & Kontoyiannis, D. P. (2003). Glucocorticoids and invasive fungal infections. Lancet, 362(9398), 1828-1838. https://doi.org/10.1016/S0140-6736(03)14904-5
- Fishman, J. A., & Rubin, R. H. (1998). Infection in organ-transplant recipients. New England Journal of Medicine, 338(24), 1741-1751. https://doi.org/10.1056/NEJM199806113382407
- Calandra, T., & Cohen, J. (2005). The international sepsis forum consensus conference on definitions of infection in the intensive care unit. Critical Care Medicine, 33(7), 1538-1548. https://doi.org/10.1097/01.CCM.0000168253.91200.83
- Kroger, A. T., Duchin, J., & Vázquez, M. (2018). General best practice guidelines for immunization: Best practices guidance of the Advisory Committee on Immunization Practices (ACIP). Centers for Disease Control and Prevention.
- Greenberg, R. N. (1991). Overview of patient compliance with medication dosing: A literature review. Clinical Therapeutics, 6(5), 592-599.
- Peckett, A. J., Wright, D. C., & Riddell, M. C. (2011). The effects of glucocorticoids on adipose tissue lipid metabolism. Metabolism, 60(11), 1500-1510. https://doi.org/10.1016/j.metabol.2011.06.012
- Tataranni, P. A., Larson, D. E., Snitker, S., Young, J. B., Flatt, J. P., & Ravussin, E. (1996). Effects of glucocorticoids on energy metabolism and food intake in humans. American Journal of Physiology-Endocrinology and Metabolism, 271(2), E317-E325. https://doi.org/10.1152/ajpendo.1996.271.2.E317
- Whitworth, J. A. (1987). Studies on the mechanisms of glucocorticoid hypertension in humans. Blood Pressure, 6(1), 24-28. https://doi.org/10.3109/08037059709086445
- Rockall, A. G., Sohaib, S. A., Evans, D., Kaltsas, G., Isidori, A. M., Monson, J. P., … & Grossman, A. B. (2003). Hepatic steatosis in Cushing’s syndrome: A radiological assessment using computed tomography. European Journal of Endocrinology, 149(6), 543-548. https://doi.org/10.1530/eje.0.1490543
- McMahon, M., Gerich, J., & Rizza, R. (1988). Effects of glucocorticoids on carbohydrate metabolism. Diabetes/Metabolism Research and Reviews, 4(1), 17-30. https://doi.org/10.1002/dmr.5610040105
- Geer, E. B., Islam, J., & Buettner, C. (2014). Mechanisms of glucocorticoid-induced insulin resistance: Focus on adipose tissue function and lipid metabolism. Endocrinology and Metabolism Clinics of North America, 43(1), 75-102. https://doi.org/10.1016/j.ecl.2013.10.005
- Silva, J. A., Jacobs, J. W., Kirwan, J. R., Boers, M., Saag, K. G., Inês, L. B., … & Bijlsma, J. W. (2006). Safety of low dose glucocorticoid treatment in rheumatoid arthritis: Published evidence and prospective trial data. Annals of the Rheumatic Diseases, 65(3), 285-293. https://doi.org/10.1136/ard.2005.038638
- Oray, M., Abu Samra, K., Ebrahimiadib, N., Meese, H., & Foster, C. S. (2016). Long-term side effects of glucocorticoids. Expert Opinion on Drug Safety, 15(4), 457-465. https://doi.org/10.1517/14740338.2016.1140743
- Huscher, D., Thiele, K., Gromnica-Ihle, E., Hein, G., Demary, W., Dreher, R., … & Zink, A. (2009). Dose-related patterns of glucocorticoid-induced side effects. Annals of the Rheumatic Diseases, 68(7), 1119-1124. https://doi.org/10.1136/ard.2008.092163
- van Staa, T. P., Leufkens, H. G., Abenhaim, L., Begaud, B., Zhang, B., & Cooper, C. (2000). Use of oral corticosteroids in the United Kingdom. QJM, 93(2), 105-111. https://doi.org/10.1093/qjmed/93.2.105
- Lansang, M. C., & Hustak, L. K. (2011). Glucocorticoid-induced diabetes and adrenal suppression: How to detect and manage them. Cleveland Clinic Journal of Medicine, 78(11), 748-756. https://doi.org/10.3949/ccjm.78a.10180
- Canalis, E., Mazziotti, G., Giustina, A., & Bilezikian, J. P. (2007). Glucocorticoid-induced osteoporosis: Pathophysiology and therapy. Osteoporosis International, 18(10), 1319-1328. https://doi.org/10.1007/s00198-007-0394-0
- Sarnes, E., Crofford, L., Watson, M., Dennis, G., Kan, H., & Bass, D. (2011). Incidence and US costs of corticosteroid-associated adverse events: A systematic literature review. Clinical Therapeutics, 33(10), 1413-1432. https://doi.org/10.1016/j.clinthera.2011.09.009
- McDonough, A. K., Curtis, J. R., & Saag, K. G. (2008). The epidemiology of glucocorticoid-associated adverse events. Current Opinion in Rheumatology, 20(2), 131-137. https://doi.org/10.1097/BOR.0b013e3282f51031
How long does it take for prednisone to start working?
The onset of action for prednisone may vary depending on the condition being treated and individual patient factors.[152]
Some patients may experience initial effects within hours of administration, particularly for acute inflammatory conditions.[153]
However, maximum therapeutic effects may not be apparent for several days to weeks, especially for chronic inflammatory or autoimmune conditions.[154]
The medication’s anti-inflammatory effects may begin relatively quickly, but tissue healing and resolution of chronic inflammation may require longer periods of treatment.[155]
Patients should be counseled that while some improvement may be noticed early in treatment, optimal therapeutic benefit may take time to develop.[156]
Can prednisone be taken with food?
Prednisone tablets may be taken with or without food, though administration with meals may help reduce potential gastrointestinal side effects.[157]
Taking the medication with food or milk may help minimize stomach irritation that some patients experience with corticosteroid therapy.[158]
The bioavailability of prednisone is generally not significantly affected by food intake, so timing with meals is primarily for comfort rather than efficacy.[159]
Patients who experience stomach upset should be advised to take their medication with food.[160]
However, consistency in administration timing, whether with or without food, may help maintain steady drug levels.[161]
What should I do if I miss a dose of prednisone?
Missing a dose of prednisone may require different approaches depending on the dosing schedule and timing of the missed dose.[162]
For once-daily dosing, if the missed dose is remembered within a few hours, it may generally be safe to take the missed dose.[163]
However, if it is nearly time for the next scheduled dose, patients should typically skip the missed dose and resume their regular schedule.[164]
Double dosing should be avoided as it may increase the risk of adverse effects.[165]
Patients receiving multiple daily doses should consult their healthcare provider for specific guidance on missed doses.[166]
For patients on long-term therapy, missing occasional doses may be less problematic than for those receiving short-term high-dose treatment.[167]
Are there any dietary restrictions while taking prednisone?
While there are no absolute dietary restrictions with prednisone, certain dietary modifications may help minimize potential side effects.[168]
Reducing sodium intake may help prevent fluid retention and blood pressure elevation that can occur with corticosteroid therapy.[169]
Adequate calcium and vitamin D intake may be important for bone health, particularly during long-term treatment.[170]
Patients may benefit from a diet rich in potassium to help counteract potential potassium loss associated with corticosteroid use.[171]
Blood glucose monitoring may be necessary for patients with diabetes, as prednisone can affect glucose control.[172]
Limiting simple carbohydrates and maintaining a balanced diet may help manage weight gain that commonly occurs with corticosteroid therapy.[173]
Can I drink alcohol while taking prednisone?
Alcohol consumption during prednisone therapy may increase the risk of certain adverse effects and should generally be limited or avoided.[174]
Both alcohol and corticosteroids may increase the risk of gastrointestinal ulceration and bleeding, particularly when used together.[175]
Alcohol may also interfere with blood glucose control, which could be problematic for patients already experiencing hyperglycemic effects from prednisone.[176]
Additionally, alcohol may affect immune function and could potentially compound the immunosuppressive effects of corticosteroids.[177]
Patients should discuss alcohol use with their healthcare provider to determine appropriate guidelines based on their individual situation and treatment plan.[178]
How should prednisone be discontinued?
Prednisone should generally not be stopped abruptly, particularly after prolonged use, due to the risk of adrenal insufficiency.[179]
Gradual dose reduction allows the hypothalamic-pituitary-adrenal axis to recover and resume normal cortisol production.[180]
The tapering schedule may depend on the duration of therapy, with slower tapering required for patients who have received treatment for extended periods.[181]
A common approach involves reducing the dose by 10-25% every 1-2 weeks, though individualization may be necessary.[182]
Some patients may experience withdrawal symptoms during tapering, including fatigue, joint pain, and mood changes.[183]
Healthcare providers should monitor patients closely during the discontinuation process and may need to adjust the tapering schedule based on patient response.[184]
Can prednisone affect my mood or mental state?
Prednisone may indeed affect mood and mental state, with psychiatric side effects being relatively common.[185]
Patients may experience mood elevation, euphoria, irritability, anxiety, or depression during treatment.[186]
More severe psychiatric effects, including psychosis or severe depression, may occur but are less common.[187]
These effects may be dose-related and could occur even with short-term therapy.[188]
Patients with a history of psychiatric disorders may be at higher risk for developing mood-related side effects.[189]
Family members and caregivers should be aware of potential mood changes and report concerning symptoms to healthcare providers.[190]
In some cases, psychiatric medications or dose adjustments may be necessary to manage these effects.[191]
Will prednisone affect my ability to fight infections?
Prednisone may suppress immune system function and could reduce the body’s ability to fight infections.[192]
The immunosuppressive effects may increase susceptibility to bacterial, viral, fungal, and parasitic infections.[193]
Patients should be advised to avoid contact with individuals who have contagious illnesses when possible.[194]
Signs and symptoms of infection may be masked by the anti-inflammatory effects of corticosteroids, potentially delaying diagnosis and treatment.[195]
Live vaccines should generally be avoided during prednisone therapy due to the risk of vaccine-induced infections.[196]
Patients should promptly report any signs of infection, such as fever, sore throat, or unusual fatigue, to their healthcare provider.[197]
Can prednisone cause weight gain?
Weight gain is a common side effect of prednisone therapy that may result from multiple mechanisms.[198]
The medication may increase appetite, leading to increased caloric intake.[199]
Fluid retention due to sodium retention may also contribute to weight gain.[200]
Additionally, prednisone may cause redistribution of body fat, leading to increased abdominal weight and characteristic cushingoid features.[201]
Metabolic changes, including altered glucose metabolism, may also play a role in weight gain.[202]
Patients should be counseled about the potential for weight gain and encouraged to maintain a healthy diet and exercise routine when possible.[203]
Weight monitoring during therapy may help identify significant changes that might require intervention.[204]
How often will I need blood tests while taking prednisone?
The frequency of laboratory monitoring during prednisone therapy may depend on the dose, duration of treatment, and individual patient factors.[205]
Baseline laboratory studies may include complete blood count, comprehensive metabolic panel, and glucose levels.[206]
During therapy, monitoring may include assessment of electrolytes, glucose, and markers of bone metabolism.[207]
Patients with diabetes may require more frequent glucose monitoring due to prednisone’s hyperglycemic effects.[208]
Long-term therapy may warrant periodic assessment of bone density and cardiovascular risk factors.[209]
The specific monitoring schedule should be individualized based on the patient’s overall health status and risk factors.[210]
Healthcare providers will determine the appropriate frequency and type of monitoring based on clinical circumstances.[211]
Disclaimer:
This product information is provided for educational purposes only and is not intended to replace professional medical advice, diagnosis, or treatment. Always consult with a qualified healthcare provider before starting, stopping, or modifying any medication regimen. The information presented herein does not constitute medical advice and should not be used as a substitute for consultation with a licensed healthcare professional. Individual patient responses to medications may vary significantly, and treatment decisions should always be made in consultation with appropriate medical supervision. This document does not imply endorsement, recommendation, or guarantee of safety or efficacy for any particular use. Healthcare providers should refer to current prescribing information and clinical guidelines when making treatment decisions.
503A vs 503B
- 503A pharmacies compound products for specific patients whose prescriptions are sent by their healthcare provider.
- 503B outsourcing facilities compound products on a larger scale (bulk amounts) for healthcare providers to have on hand and administer to patients in their offices.
Frequently asked questions
Our team of experts has the answers you're looking for.
A clinical pharmacist cannot recommend a specific doctor. Because we are licensed in all 50 states*, we can accept prescriptions from many licensed prescribers if the prescription is written within their scope of practice and with a valid patient-practitioner relationship.
*Licensing is subject to change.
Each injectable IV product will have the osmolarity listed on the label located on the vial.

Given the vastness and uniqueness of individualized compounded formulations, it is impossible to list every potential compound we offer. To inquire if we currently carry or can compound your prescription, please fill out the form located on our Contact page or call us at (877) 562-8577.
We source all our medications and active pharmaceutical ingredients from FDA-registered suppliers and manufacturers.

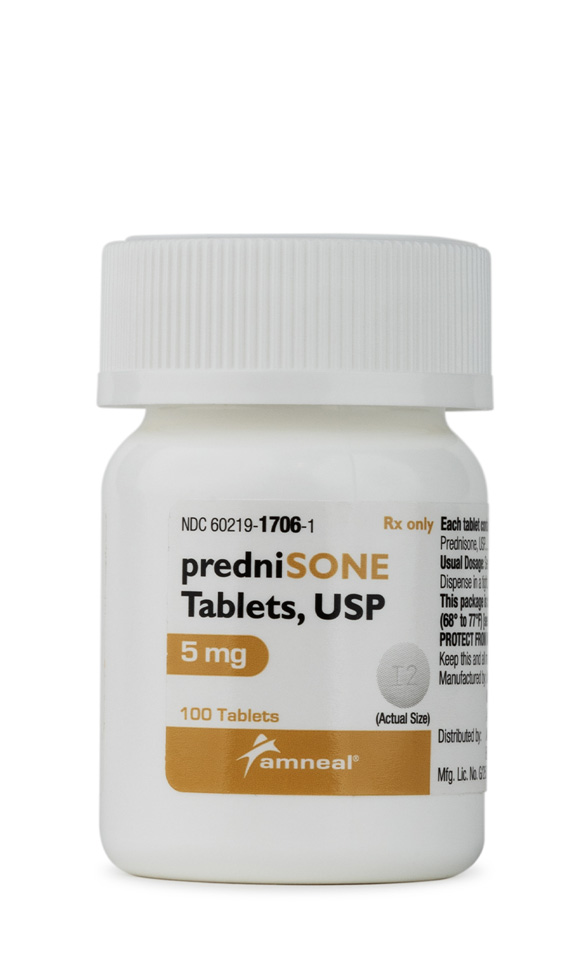
 Dexamethasone Injection
Dexamethasone Injection Ketorolac Tromethamine Injection
Ketorolac Tromethamine Injection Hydrocortisone Tablets
Hydrocortisone Tablets Ascorbic Acid (Vitamin C) Injection
Ascorbic Acid (Vitamin C) Injection Calcium Gluconate Injection
Calcium Gluconate Injection Diphenhydramine Injection
Diphenhydramine Injection Vitamin B-Complex Injection
Vitamin B-Complex Injection Magnesium Chloride Injection
Magnesium Chloride Injection