Cyanocobalamin (Vitamin B12) Injection
† commercial product
This product is available through both our 503A Compounding Pharmacy and our 503B Outsourcing Facility, ensuring versatile solutions tailored to meet your needs. If you do not have an account, please contact us.
Product Overview
Cyanocobalamin (vitamin B12) injection is a sterile, prescription-only parenteral formulation of a water-soluble cobalamin analogue that serves as the principal therapeutic option when oral absorption of the vitamin is impaired or when rapid repletion is clinically required. It is dispensed in amber multidose vials containing 1,000 mcg/mL and compounded for use under either section 503A or 503B, allowing clinicians to individualize therapy for deficiency states ranging from pernicious anemia to postsurgical malabsorption.[1]
Because intrinsic-factor-dependent uptake in the distal ileum can be absent in conditions such as autoimmune gastritis, pancreatic insufficiency, or bariatric surgery, the intramuscular or subcutaneous route bypasses gastrointestinal barriers and reliably restores serum concentrations. The product is likewise indicated for increased physiologic demand (pregnancy, thyrotoxicosis, chronic hemorrhage), for drug-induced depletion, and for select neurologic or hematologic disorders in which B12 plays a pivotal metabolic role.[2]
Pharmacokinetic studies demonstrate rapid systemic distribution with peak plasma levels occurring within 30-60 minutes of intramuscular injection, followed by extensive hepatic storage and enterohepatic recycling; roughly 50-98 % of an administered dose is renally excreted unmetabolized, underscoring the low intrinsic toxicity.[3]
Initial treatment of pernicious anemia typically consists of 1,000 mcg intramuscularly daily for one week, then weekly for four weeks, transitioning to 1,000 mcg monthly for life;
alternative protocols (e.g., 100 mcg every other day) may be selected based on clinical response and laboratory indices.[20]
For nutritional deficiency without intrinsic-factor loss, shorter courses-such as 30-100 mcg intramuscularly daily for five to ten days followed by 100-200 mcg monthly-are often sufficient; pediatric and intranasal regimens are detailed in specialized dosing references and should be individualized.[21]
Within cells, cyanocobalamin is converted to methylcobalamin and 5- deoxyadenosylcobalamin, coenzymes that participate respectively in homocysteine remethylation to methionine and in the isomerization of methylmalonyl-CoA to succinyl CoA, reactions essential for DNA synthesis, erythropoiesis, and myelin maintenance.[5]
Disruption of these pathways leads to accumulation of homocysteine and methylmalonic acid, metabolic signatures linked to megaloblastic anemia and demyelinating neuropathy; repletion reverses hematologic symptoms within days and neurologic sequelae over weeks, reflecting differential tissue turnover.[6]
After parenteral delivery, cyanocobalamin binds transcobalamin II, permitting receptor mediated endocytosis into virtually all nucleated cells; intracellular half-life is prolonged because of lysosomal release and cytosolic resequestration, creating a depot effect that supports monthly maintenance dosing for chronic deficiencies.[7]
Population pharmacokinetic modeling reveals a biphasic disposition (distribution half-life 15-30 minutes; terminal half-life 20-50 minutes) with a steady-state volume approximating total extracellular fluid, findings that justify loading schedules followed by less frequent injections once tissue stores exceed 2 mg.[8]
Absolute contraindications include prior hypersensitivity to cyanocobalamin, cobalt, or formulation excipients; anaphylaxis, though rare, necessitates immediate discontinuation and emergency management.[9]
Visual disturbances have been reported in patients with Leber hereditary optic neuropathy, and rapid B12 administration may precipitate optic nerve atrophy in this genetically predisposed population, mandating extreme caution or alternative therapy.[10]
Relative contraindications encompass early polycythemia vera, tobacco amblyopia, and concurrent sensitivity to benzyl alcohol used as a preservative; in such settings the risk benefit ratio should be reassessed, and preservative-free preparations considered.[11]
Long-term metformin therapy reduces calcium-dependent intrinsic-factor binding, lowering B12 absorption and potentially necessitating higher or more frequent parenteral doses; periodic monitoring is prudent.[12]
Gastric acid-suppressing agents (proton-pump inhibitors and H₂-receptor antagonists) diminish dietary B12 liberation from protein complexes, a pharmacodynamic interaction that can exacerbate underlying deficiency.[13]
Chloramphenicol has been shown to blunt reticulocyte response to cyanocobalamin, while ethanol, colchicine, and nitrous oxide interfere with cobalamin metabolism or utilization, each warranting clinician awareness when evaluating suboptimal treatment outcomes.[14]
The injection is generally well tolerated; common reactions include transient pain, erythema, or pruritus at the administration site, along with mild diarrhea, headache, or nausea in a minority of recipients.[15]
Serious but infrequent events-such as angioedema, bronchospasm, hypotension, or urticaria-reflect IgE-mediated hypersensitivity and may require desensitization or substitution with hydroxocobalamin.[16]
Although cyanocobalamin lacks a defined upper-toxicity threshold due to its water solubility and renal elimination, exceptionally high cumulative doses have been temporally associated with acneiform eruptions, altered potassium homeostasis, and theoretical thrombotic risk, emphasizing the importance of evidence-based dosing.[17]
Physiologic B12 requirements increase during gestation, and parenteral cyanocobalamin is considered appropriate when dietary intake or intestinal absorption is inadequate; limited human data reveal no teratogenicity at therapeutic doses, yet clinicians are advised to employ the lowest effective regimen.[18]
Lactation studies demonstrate that maternal injections modestly elevate breast-milk B12 concentrations without adverse infant outcomes, supporting compatibility with breastfeeding while underscoring the need for maternal repletion to prevent neonatal deficiency.[19]
Cyanocobalamin vials should be stored at controlled room temperature (15 - 25 °C), protected from excessive light and humidity, and kept in their original packaging to preserve potency; freezing must be avoided to prevent crystal formation and loss of sterility assurance.[22]
Refrigeration is not strictly required but may prolong shelf life if temperature excursions are common; when cooled, vials must be allowed to reach ambient temperature before use to minimize syringe air bubble formation and injection discomfort.[23]
- Medicine..com. (2020, February 17). Cyanocobalamin: Dosage, mechanism/onset of action, half-life. https://www.medicine.com/drug/cyanocobalamin/hcp
- Mayo Clinic. (2025). Cyanocobalamin (intramuscular route). https://www.mayoclinic.org/drugs-supplements/cyanocobalamin-intramuscular-route/description/drg-20137833
- Allen, L. H. (2022). Assessing vitamin B12 absorption and bioavailability: Read the label. American Journal of Clinical Nutrition, 115(4), 787-789. https://doi.org/10.1093/ajcn/nqac004
- U.S. Food and Drug Administration. (2024). CYANOCOBALAMIN injection, USP [DailyMed label]. https://fda.report/DailyMed/a66eb3c4-3e1d-4d49-b963-4fa2334cc9b6
- Khanduri, U., & Sharma, A. (2020). Deficiency of vitamin B12 and its relation with neurological disorders: A review. Basic and Applied Zoology, 81, 110. https://doi.org/10.1186/s41936-020-00148-0
- Singh, A. et al. (2025). Vitamin B12 deficiency and cognitive impairment: A comprehensive review. Clinical Nutrition Open Science, 41, 100065. https://doi.org/10.1016/j.nutos.2025.100065
- Levy, R. et al. (2023). Pharmacokinetics of high doses of cyanocobalamin administered intramuscularly. Journal of Clinical Pharmacology, 63(12), 1121-1130. https://cris.ariel.ac.il/en/publications/pharmacokinetics-of-high-doses-of-cyanocobalamin-administered-by–3
- UK Medicines and Healthcare Products Regulatory Agency. (2019). Clinical overview: Cyanocobalamin 1 mg film-coated tablet. https://assets.publishing.service.gov.uk/media/61e98bae8fa8f50594b59420/FOI_21-1119-2.pdf
- Drugs..com. (2024). Cyanocobalamin injection: Uses, side effects & warnings. https://www.drugs.com/mtm/cyanocobalamin-injection.html
- RxList. (2022). Cyanocobalamin (cyanocobalamin): Side effects, uses, dosage. https://www.rxlist.com/cyanocobalamin-drug.htm
- Memorial Sloan Kettering Cancer Center. (2025). Cyanocobalamin: Patient and caregiver education. https://www.mskcc.org/cancer-care/patient-education/medications/adult/cyanocobalamin
- EBM Consult. (2023). Medications known to decrease vitamin B12 levels. https://www.ebmconsult.com/articles/vitamin-b12-medication-interactions-lower-levels
- WebMD. (2024). Vitamin B-12 injection: Patient information. https://www.webmd.com/drugs/2/drug-6550/vitamin-b-12-injection/details
- EatingWell. (2025). The best time of day to take vitamin B12 for maximum-absorption. https://www.eatingwell.com/the-best-time-of-day-to-take-vitamin-b12-for-maximum absorption-8782337
- Drugs..com. (2025). Cyanocobalamin side effects: Consumer information. https://www.drugs.com/sfx/cyanocobalamin-side-effects.html
- Reactions Weekly. (2025). Hypersensitivity reaction to cyanocobalamin. https://link.springer.com/article/10.1007/s40278-025-84053-5
- SingleCare. (2023). Too much B12: Signs, causes, and how to manage it. https://www.singlecare.com/conditions/too-much-B12-symptoms
- Drugs..com. (2024). Cyanocobalamin use during pregnancy. https://www.drugs.com/pregnancy/cyanocobalamin.html
- National Health Service. (2024). Pregnancy, breastfeeding and cyanocobalamin. https://www.nhs.uk/medicines/cyanocobalamin/pregnancy-breastfeeding-and-fertility-while-taking-cyanocobalamin/
- Drugs..com. (2023). Cyanocobalamin dosage guide. https://www.drugs.com/dosage/cyanocobalamin.html
- Medscape. (2025). Vitamin B12 (cyanocobalamin) dosing, indications, interactions. https://reference.medscape.com/drug/vitamin-b12-nascobal-cyanocobalamin-344418
- B12 Vitamin Store. (2024). Storage instructions for vitamin B12. https://b12vitaminstore.com/storage-instructions-b12-cyanocobalamin
- ApplianceUpdate. (2025). Does cyanocobalamin need to be refrigerated? https://applianceupdate.com/does-cyanocobalamin-have-to-be-refrigerated/
- Times of India. (2025). Vitamin B12 and brain fog. https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/vitamin-b12-and-brain-fog-the-connection-that-is-more-common-than-you-think/photostory/122116448.cms
- Medicover Hospitals. (2025). Neurological impact of vitamin B12 deficiency. https://www.medicoverhospitals.in/articles/neurological-effects-of-vitamin-b12-deficiency
- Wellwisp. (2024). How do you store B12 injections? https://wellwisp.com/how-do-you store-b12-injections/
- Pernicious Anemia Relief. (2024). Vitamin B12 overdose & toxicity. https://perniciousanemia.org/b12/toxicity/
- Centers for Disease Control and Prevention. (2023). Vitamin B12 and breastfeeding.https://www.cdc.gov/breastfeeding-special-circumstances/hcp/diet micronutrients/vitamin-b12.htm
- Drugs..com. (2024). Vitamin B12 use while breastfeeding. https://www.drugs.com/breastfeeding/vitamin-b-sub-12-sub.html
- NDrugs. (2023). Cyanocobalamin injection: Side effects and contraindications. https://www.ndrugs.com/?s=cyanocobalamin++injection&t=side+effects
- LiveStrong. (2023). Normal intramuscular dose of vitamin B12.
- https://www.livestrong.com/article/474571-what-is-the-normal-intramuscular-dose-of vitamin-b12/
- PedMed. (2024). Cyanocobalamin dosing in children. https://pedmed.org/pedshowdrug.php?drugID=102
- MPR. (2025). Cyanocobalamin injection: Prescription & dosage information. https://www.empr.com/drug/cyanocobalamin-injection/
Can B12 injections improve “brain fog” in otherwise healthy adults?
Cognitive benefits are primarily documented in deficient individuals; routine use in replete patients has limited evidence.[24]
How soon should neurologic symptoms begin to resolve after starting therapy?
Paresthesias and gait disturbances may begin to improve within two weeks, although full recovery can take several months.[25]
Is overdose dangerous?
No lethal dose has been identified; excess is renally excreted, though very high levels can cause dermatologic or metabolic effects.[27]
Does maternal therapy affect breast-feeding infants?
Adequate maternal B12 supports infant nutrition, and standard doses are considered safe during lactation.[28]
Should nursing mothers take additional oral B12 with injections?
Supplementation is unnecessary if parenteral therapy repletes serum levels, but individual assessment is advised.[29]
Why do some patients still feel fatigued after injections?
Concomitant deficiencies (iron, folate), chronic illness, or drug interactions may blunt clinical response and warrant further evaluation.[30]
What is the typical intramuscular maintenance dose?
Many adults remain stable on 1,000 mcg once monthly, though dose intervals can be adjusted according to serum markers.[31]
Are pediatric doses the same as adult doses?
Children generally require weight-adjusted regimens, often 100 mcg monthly after initial loading; specialist guidance is essential.[32]
Can injections be self-administered?
With proper aseptic training and follow-up, select patients may self-inject subcutaneously, enhancing adherence and convenience.[33]
Administration Instructions
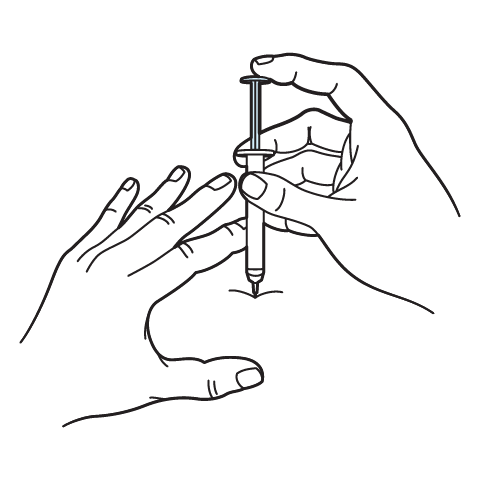
Intramuscular Injection Instructions
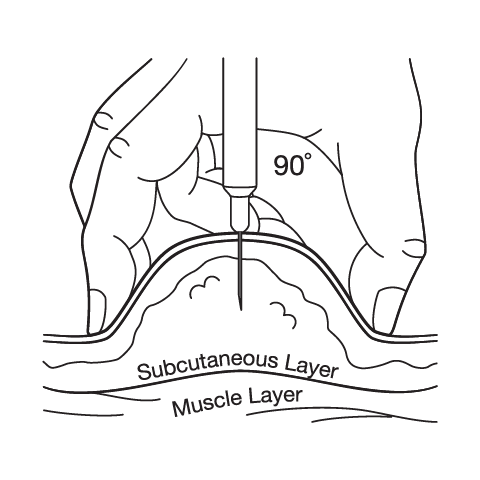
Subcutaneous Injection Instructions
503A vs 503B
- 503A pharmacies compound products for specific patients whose prescriptions are sent by their healthcare provider.
- 503B outsourcing facilities compound products on a larger scale (bulk amounts) for healthcare providers to have on hand and administer to patients in their offices.
Frequently asked questions
Our team of experts has the answers you're looking for.
A clinical pharmacist cannot recommend a specific doctor. Because we are licensed in all 50 states*, we can accept prescriptions from many licensed prescribers if the prescription is written within their scope of practice and with a valid patient-practitioner relationship.
*Licensing is subject to change.
Each injectable IV product will have the osmolarity listed on the label located on the vial.

Given the vastness and uniqueness of individualized compounded formulations, it is impossible to list every potential compound we offer. To inquire if we currently carry or can compound your prescription, please fill out the form located on our Contact page or call us at (877) 562-8577.
We source all our medications and active pharmaceutical ingredients from FDA-registered suppliers and manufacturers.



 Hydroxocobalamin (Vitamin B12) Injection
Hydroxocobalamin (Vitamin B12) Injection Methylcobalamin Injection (Vitamin B12)
Methylcobalamin Injection (Vitamin B12) Vitamin B-Complex Injection
Vitamin B-Complex Injection Ascorbic Acid (Vitamin C) Injection
Ascorbic Acid (Vitamin C) Injection Glutathione Injection
Glutathione Injection NAD+ Injection (Lyo)
NAD+ Injection (Lyo) Zinc Sulfate Injection
Zinc Sulfate Injection